Γ-Hydroxybutyraldehyde
Γ-Hydroxybutyraldehyde (also known as 4-hydroxybutanal) is an organic compound with the formula C₄H₈O₂. It is a colorless liquid that is an intermediate in the metabolism of γ-Hydroxybutyric acid (GHB). This compound is of interest in both biochemical research and industrial applications.
Structure and Properties[edit]
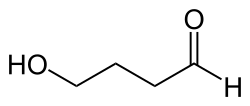
Γ-Hydroxybutyraldehyde is an aldehyde with a hydroxyl group on the fourth carbon of the butanal chain. This structure gives it both aldehydic and alcoholic properties, making it reactive in various chemical processes. The presence of the hydroxyl group increases its solubility in water compared to other aldehydes.
Synthesis[edit]
Γ-Hydroxybutyraldehyde can be synthesized through the oxidation of 1,4-butanediol or by the reduction of succinic acid. In laboratory settings, it is often prepared by the partial oxidation of 1,4-butanediol using mild oxidizing agents.
Biological Role[edit]
In biological systems, Γ-hydroxybutyraldehyde is an intermediate in the metabolic pathway of γ-Hydroxybutyric acid (GHB). GHB is a naturally occurring neurotransmitter and a psychoactive drug. The conversion of GHB to Γ-hydroxybutyraldehyde is catalyzed by the enzyme GHB dehydrogenase.
Metabolic Pathway[edit]
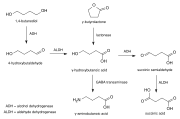
The metabolic pathway of GHB involves its conversion to Γ-hydroxybutyraldehyde, which is then further oxidized to succinic semialdehyde. This compound is subsequently converted to succinate, which enters the citric acid cycle. This pathway highlights the role of Γ-hydroxybutyraldehyde as a key intermediate in energy metabolism.
Applications[edit]
Γ-Hydroxybutyraldehyde is used in the synthesis of various pharmaceuticals and as a precursor in the production of polymers. Its reactivity makes it a valuable intermediate in organic synthesis, particularly in the formation of cyclic compounds and in aldol reactions.
Safety and Handling[edit]
As with many aldehydes, Γ-hydroxybutyraldehyde should be handled with care. It is a volatile compound that can cause irritation to the skin, eyes, and respiratory tract. Proper safety equipment, such as gloves and goggles, should be used when handling this chemical.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian