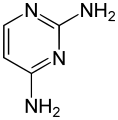
2,4-Diaminopyrimidine
2,4-Diaminopyrimidine is an organic compound with the formula C4H6N4. It is one of the isomers of diaminopyrimidine, a type of pyrimidine derivative. Pyrimidines are aromatic heterocyclic organic compounds similar to benzene and pyridine, containing two nitrogen atoms at positions 1 and 3 of the six-member ring.
Structure and Properties[edit]
2,4-Diaminopyrimidine is a white crystalline solid at room temperature. It is soluble in water and most organic solvents. The compound has two amino groups (-NH2) attached to the carbon atoms at the 2nd and 4th positions of the pyrimidine ring. This structure is responsible for its basicity and ability to form salts with acids.
Synthesis[edit]
The synthesis of 2,4-Diaminopyrimidine typically involves the reaction of guanidine with cyanoacetic acid in the presence of a base. The resulting intermediate is then cyclized to form the pyrimidine ring.
Applications[edit]
2,4-Diaminopyrimidine is used as a building block in the synthesis of various pharmaceuticals and agrochemicals. It is also used in the production of dyes and pigments, and as a corrosion inhibitor in the metal industry.
Safety and Toxicity[edit]
Like many other pyrimidine derivatives, 2,4-Diaminopyrimidine is potentially toxic and should be handled with care. It may cause irritation to the skin and eyes, and prolonged exposure may lead to more serious health effects.
See Also[edit]
2,4-Diaminopyrimidine gallery[edit]
-
2,4-Diaminopyrimidine
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian