3-Chloropropanoic acid
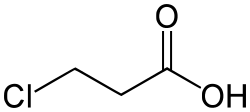
3-Chloropropanoic acid is an organic compound with the formula ClCH2CH2COOH. It is a chlorocarboxylic acid that is a derivative of propanoic acid with a chlorine atom replacing a hydrogen atom on the second carbon of the chain. This compound is important in the field of organic chemistry due to its reactivity and role in various chemical syntheses.
Properties[edit]
3-Chloropropanoic acid is a colorless liquid at room temperature and has a pungent odor. It is soluble in water, ethanol, and diethyl ether. The presence of the chlorine atom significantly affects the acid's physical and chemical properties, making it more reactive than propanoic acid. The chlorine atom is electron-withdrawing, which increases the acidity of the carboxylic acid group. Therefore, 3-Chloropropanoic acid has a lower pKa than propanoic acid, making it a stronger acid.
Synthesis[edit]
3-Chloropropanoic acid can be synthesized through the chlorination of propanoic acid using agents such as thionyl chloride, phosphorus trichloride, or phosphorus pentachloride. Another method involves the hydrolysis of 3-chloropropionitrile, which is derived from the reaction of acrylonitrile with hydrogen chloride.
Applications[edit]
In organic synthesis, 3-Chloropropanoic acid is used as a building block for the production of various pharmaceuticals and agrochemicals. Its reactivity allows for the introduction of a chlorine atom into molecules, which can be further transformed through reactions such as nucleophilic substitution or elimination. It is also used in the synthesis of esters, amides, and other functionalized derivatives, which have applications in different industrial sectors.
Safety[edit]
3-Chloropropanoic acid is corrosive and can cause severe burns upon contact with skin or eyes. It should be handled with appropriate safety precautions, including the use of personal protective equipment (PPE) such as gloves and safety glasses. Proper ventilation is also important to avoid inhalation of fumes.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
