4-Caffeoyl-1,5-quinide
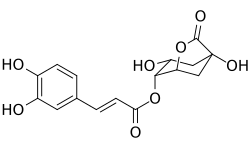
Chemical compound
| 4-Caffeoyl-1,5-quinide | |
|---|---|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider ID | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| Properties | |
| Chemical Formula | |
| Molar Mass | |
| Appearance | |
| Density | |
| Melting Point | |
| Boiling Point | |
| Hazards | |
| GHS Pictograms | [[File:|50px]] |
| GHS Signal Word | |
| GHS Hazard Statements | |
| NFPA 704 | [[File:|50px]] |
| References | |
4-Caffeoyl-1,5-quinide is a chemical compound that belongs to the class of quinides. It is a derivative of caffeic acid and is structurally related to chlorogenic acid. This compound is of interest in the field of phytochemistry and has been studied for its potential biological activity.
Structure and Properties[edit]
4-Caffeoyl-1,5-quinide is characterized by the presence of a caffeoyl group attached to a quinide structure. The molecular structure includes a phenolic ring, which is responsible for its antioxidant properties. The compound is typically studied using techniques such as NMR spectroscopy, mass spectrometry, and chromatography.
Sources[edit]
This compound can be found in various plant species, particularly those that are rich in phenolic compounds. It is often identified in the context of coffee research, as it is a derivative of compounds found in coffee beans.
Biological Activity[edit]
Research has indicated that 4-Caffeoyl-1,5-quinide may exhibit several biological activities, including antioxidant, anti-inflammatory, and antimicrobial effects. These properties make it a compound of interest for potential therapeutic applications.
Synthesis[edit]
The synthesis of 4-Caffeoyl-1,5-quinide can be achieved through various chemical reactions involving caffeic acid and related compounds. The process typically involves esterification and cyclization reactions.
Applications[edit]
Due to its potential biological activity, 4-Caffeoyl-1,5-quinide is studied for its possible use in pharmaceuticals, nutraceuticals, and cosmetics. Its antioxidant properties are particularly valued in the development of products aimed at reducing oxidative stress.
See also[edit]
References[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian