AMG-41
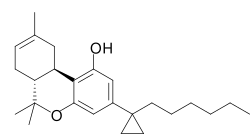
Experimental antiretroviral drug
| AMG-41
|
|---|
AMG-41 is an experimental antiretroviral drug being developed for the treatment of HIV/AIDS. It belongs to a class of medications known as entry inhibitors, which work by preventing the HIV virus from entering human cells.
Mechanism of Action[edit]
AMG-41 functions as an entry inhibitor by targeting the gp120 protein on the surface of the HIV virus. This protein is essential for the virus to bind to the CD4 receptors on T cells, which are a type of white blood cell crucial for the immune system. By blocking this interaction, AMG-41 prevents the virus from entering and infecting the cells.
Development and Clinical Trials[edit]
AMG-41 is currently in the early stages of clinical development. Initial studies have shown promise in its ability to reduce viral load in patients with HIV. Further clinical trials are ongoing to determine its efficacy and safety profile.
Potential Benefits[edit]
The development of AMG-41 could provide a new option for patients who have developed resistance to existing antiretroviral therapies. Its unique mechanism of action as an entry inhibitor distinguishes it from other classes of antiretroviral drugs, such as reverse transcriptase inhibitors and protease inhibitors.
Side Effects[edit]
As AMG-41 is still under investigation, the full range of potential side effects is not yet known. However, common side effects associated with entry inhibitors may include nausea, fatigue, and headache.
See Also[edit]
References[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian