Acetone cyanohydrin
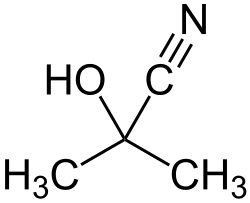
Acetone cyanohydrin is an organic compound with the formula (CH₃)₂C(OH)CN. It is a colorless liquid, although it is usually encountered as a pale yellow liquid due to impurities. It is classified as a cyanohydrin, which is a functional group consisting of a hydroxyl group and a nitrile group attached to the same carbon atom.
Synthesis[edit]
Acetone cyanohydrin is produced by the reaction of acetone with hydrogen cyanide (HCN): Failed to parse (syntax error): {\displaystyle (CH₃)₂CO + HCN → (CH₃)₂C(OH)CN } This reaction is typically catalyzed by a base such as sodium hydroxide or potassium hydroxide.
Applications[edit]
Acetone cyanohydrin is primarily used as an intermediate in the production of methyl methacrylate (MMA), which is a monomer used to produce polymethyl methacrylate (PMMA), a transparent thermoplastic often used as a lightweight or shatter-resistant alternative to glass. The conversion of acetone cyanohydrin to MMA involves several steps, including hydrolysis and esterification.
Safety[edit]
Acetone cyanohydrin is highly toxic and can release hydrogen cyanide upon decomposition. It is a potent inhibitor of cellular respiration, and exposure can lead to symptoms such as headache, dizziness, and respiratory distress. Proper handling and storage are essential to prevent accidental exposure.
Related Compounds[edit]
See Also[edit]
References[edit]
External Links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian