Oxime
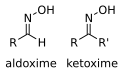
Oxime is a functional group or chemical compound containing the oxygen and nitrogen atoms connected by a double bond. The general formula for an oxime is R1R2C=NOH, where R1 is an organic side chain and R2 may be hydrogen, forming an aldoxime, or another organic group, forming a ketoxime. Oximes are usually generated by the reaction of hydroxylamine with aldehydes or ketones.
History[edit]
The oxime functional group was first synthesized in 1860 by the Russian chemist Alexander Butlerov. He produced it by reacting acetone with hydroxylamine.
Synthesis[edit]
Oximes are synthesized through the reaction of an aldehyde or ketone with hydroxylamine. The reaction is catalyzed by acid. The general formula for this reaction is R2C=O + NH2OH → R2C=NOH + H2O, where R is an organic side chain.
Properties[edit]
Oximes have a double bond between the nitrogen and oxygen atoms, and a single bond between the nitrogen atom and a hydrogen atom. They are polar compounds, due to the electronegativity difference between the oxygen and nitrogen atoms. Oximes are also capable of hydrogen bonding, which makes them soluble in water.
Applications[edit]
Oximes are used in organic chemistry as protective groups for aldehydes and ketones, and as reagents in the preparation of amines. They are also used in the medical field for the treatment of certain types of poisoning.
See also[edit]
References[edit]
Oxime[edit]
-
General structure of oximes
-
Ponzio Synthesis
-
Tiemann rearrangement
-
Caprolactam synthesis
-
Oxime
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian