Amastatin
An inhibitor of aminopeptidases
| Amastatin | |
|---|---|
|
|
Amastatin is a potent inhibitor of aminopeptidases, a class of enzymes that catalyze the cleavage of amino acids from the amino terminus of protein or peptide substrates. It is primarily used in biochemical research to study the role of aminopeptidases in various biological processes.
Structure and Properties[edit]
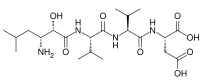
Amastatin is a naturally occurring peptide that was originally isolated from the culture broth of the bacterium Streptomyces species. It is a small molecule with a specific structure that allows it to bind effectively to the active site of aminopeptidases, thereby inhibiting their activity.
Mechanism of Action[edit]
Amastatin functions by binding to the active site of aminopeptidases, preventing the enzyme from interacting with its natural substrates. This inhibition is crucial for studying the physiological roles of aminopeptidases, as it allows researchers to observe the effects of reduced enzyme activity in various biological systems.
Applications in Research[edit]
Amastatin is widely used in biochemical and pharmacological research. It is particularly useful in studies involving the regulation of peptide hormones and neuropeptides, as aminopeptidases play a significant role in the metabolism of these molecules. By inhibiting aminopeptidases, researchers can investigate the pathways and mechanisms through which these peptides exert their effects.
Biological Significance[edit]
Aminopeptidases are involved in numerous physiological processes, including the maturation and degradation of proteins, the regulation of blood pressure, and the modulation of immune responses. Amastatin, by inhibiting these enzymes, provides insights into their function and potential as therapeutic targets in various diseases.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian