Convergent synthesis
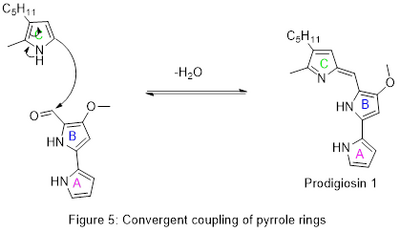
Convergent synthesis is a strategy employed in organic chemistry and medicinal chemistry for the construction of complex molecules by connecting smaller molecules together, which have been prepared separately. This approach contrasts with linear or sequential synthesis, where a molecule is constructed through a series of steps starting from a single initial molecule. Convergent synthesis is particularly useful for synthesizing large and complex molecules by combining smaller, more manageable fragments. This method can lead to significant improvements in the overall yield and efficiency of a synthetic process, as it often reduces the number of steps required to synthesize the target molecule.
The key advantage of convergent synthesis lies in its ability to minimize the multiplication of errors that is common in linear synthesis. In linear synthesis, any impurities or yield losses in early steps are propagated through to the final steps, often resulting in lower overall yields. In contrast, convergent synthesis allows for the purification of intermediate products before their final coupling, thereby improving the purity and yield of the final product.
A classic example of convergent synthesis is the synthesis of vitamin B12, which was accomplished by the chemist Robert Burns Woodward and his team. This complex natural product was synthesized from smaller, separately prepared fragments, demonstrating the power of convergent synthesis in organic chemistry.
Another important aspect of convergent synthesis is its strategic role in the field of drug discovery and development. By allowing for the rapid assembly of complex molecules from simpler precursors, convergent synthesis enables chemists to efficiently explore the chemical space around a potential drug candidate, optimizing its properties for better efficacy, safety, and pharmacokinetics.
Convergent synthesis often involves the use of protecting groups to mask reactive functional groups during the synthesis process, and coupling reactions to join the molecular fragments. The choice of coupling reaction is critical and depends on the functional groups present in the fragments to be joined. Common coupling reactions used in convergent synthesis include amide bond formation, esterification, and carbon-carbon bond forming reactions such as the Suzuki coupling.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
