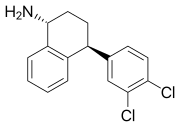
Dasotraline
Overview of the drug Dasotraline
| Dasotraline | |
|---|---|
|
|
Dasotraline is a medication that was under development for the treatment of attention deficit hyperactivity disorder (ADHD) and binge eating disorder. It is a dopamine and norepinephrine reuptake inhibitor (DNRI), which means it works by increasing the levels of these neurotransmitters in the brain.
Pharmacology[edit]
Dasotraline is classified as a dopamine reuptake inhibitor and a norepinephrine reuptake inhibitor. It has a long half-life, which allows for once-daily dosing. The drug's mechanism of action involves blocking the reuptake of dopamine and norepinephrine, thereby increasing their availability in the synaptic cleft. This action is thought to contribute to its therapeutic effects in ADHD and binge eating disorder.
Development[edit]
Dasotraline was developed by Sunovion Pharmaceuticals. It was investigated in clinical trials for the treatment of ADHD and binge eating disorder. However, as of the latest updates, the development of dasotraline for these indications has faced challenges, and its approval status remains uncertain.
Clinical Trials[edit]
Clinical trials for dasotraline have shown mixed results. In studies for ADHD, dasotraline demonstrated efficacy in reducing symptoms compared to placebo. However, concerns about side effects and the overall benefit-risk profile have been raised. In trials for binge eating disorder, dasotraline showed promise in reducing binge eating episodes.
Side Effects[edit]
Common side effects of dasotraline include insomnia, decreased appetite, and dry mouth. Some patients may experience increased heart rate and blood pressure. As with other medications affecting neurotransmitter levels, there is a potential for more serious side effects, which necessitates careful monitoring by healthcare providers.
Regulatory Status[edit]
The regulatory status of dasotraline has been complex. While it has shown potential in clinical trials, regulatory agencies have requested additional data to better understand its safety and efficacy profile. As a result, its approval for clinical use has been delayed.
Related pages[edit]
- Attention deficit hyperactivity disorder
- Binge eating disorder
- Dopamine reuptake inhibitor
- Norepinephrine reuptake inhibitor
-
Dasotraline
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian