Depsipeptide
Depsipeptide[edit]
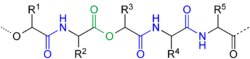
A depsipeptide is a type of peptide in which one or more of the amide bonds are replaced by ester bonds. This structural modification can significantly alter the chemical and biological properties of the peptide, making depsipeptides a subject of interest in biochemistry and pharmacology.
Structure[edit]
Depsipeptides are characterized by the presence of both amide and ester linkages within the same molecule. The ester bond is typically formed between the carboxyl group of one amino acid and the hydroxyl group of another, replacing the usual amide bond found in standard peptides. This modification can occur at one or multiple positions along the peptide chain.
Synthesis[edit]
The synthesis of depsipeptides can be achieved through various synthetic methods. One common approach is the solid-phase peptide synthesis (SPPS), which allows for the incorporation of ester bonds at specific sites. Alternatively, depsipeptides can be synthesized using enzymatic synthesis methods, which can offer greater selectivity and efficiency.
Biological Activity[edit]
Depsipeptides are known for their diverse range of biological activities. They have been found to exhibit antibiotic, antifungal, anticancer, and immunosuppressive properties. The unique structure of depsipeptides allows them to interact with biological targets in ways that are distinct from traditional peptides, often leading to enhanced or novel activities.
Applications[edit]
Due to their potent biological activities, depsipeptides are of great interest in the development of new pharmaceuticals. They are being explored as potential therapeutic agents for the treatment of various diseases, including cancer, infectious diseases, and autoimmune disorders.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian