Ethyl methanesulfonate
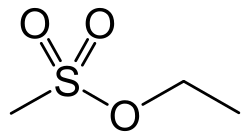
Ethyl methanesulfonate (EMS) is a chemical compound that is widely used as a mutagen in genetic research. It is an alkylating agent that induces mutations by transferring an ethyl group to DNA, which can result in point mutations.
Chemical Properties[edit]
Ethyl methanesulfonate has the chemical formula C3H8O3S and a molecular weight of 124.16 g/mol. It is a colorless liquid with a slight odor and is soluble in water and most organic solvents.
Mechanism of Action[edit]
EMS primarily induces mutations by ethylating the guanine base in DNA, which can lead to mispairing during DNA replication. This results in G:C to A:T transitions, which are a common type of point mutation. The mutagenic effects of EMS are dose-dependent and can be used to create mutant strains for genetic studies.
Applications in Research[edit]
Ethyl methanesulfonate is extensively used in mutagenesis screens to identify gene functions and to create mutant libraries. It is particularly useful in model organisms such as Drosophila melanogaster, Caenorhabditis elegans, and Arabidopsis thaliana. EMS-induced mutations have been instrumental in the discovery of numerous genes and genetic pathways.
Safety and Handling[edit]
EMS is a potent mutagen and is considered a carcinogen. Proper safety measures, including the use of personal protective equipment (PPE) and working in a fume hood, are essential when handling this chemical. It should be stored in a cool, dry place away from incompatible substances.
See Also[edit]
References[edit]
External Links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian