Iminodiacetic acid
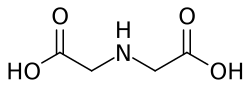
Iminodiacetic acid (IDA) is a chemical compound with the formula C4H7NO4. It is a derivative of acetic acid and is characterized by the presence of two carboxylic acid groups and one amine group. This compound is of significant interest in various fields, including biochemistry, medicine, and industrial chemistry.
Chemical Structure and Properties[edit]
Iminodiacetic acid is a white crystalline solid that is soluble in water. The molecule consists of a central nitrogen atom bonded to two acetate groups. The presence of both amine and carboxylate groups allows IDA to act as a chelating agent, forming stable complexes with metal ions.
The ability of IDA to chelate metal ions is utilized in various applications, including the purification of proteins and the removal of heavy metals from solutions. The compound's chelating properties are enhanced by the presence of the nitrogen atom, which can donate a lone pair of electrons to form coordinate bonds with metal ions.
Applications[edit]
Medical Imaging[edit]
In the field of nuclear medicine, iminodiacetic acid derivatives are used as radiopharmaceuticals for hepatobiliary imaging. These compounds, known as HIDA scans, are used to evaluate the function of the gallbladder and the biliary tract. The radiolabeled IDA compounds are taken up by the liver and excreted into the bile, allowing for the visualization of the biliary system using gamma cameras.
Industrial Uses[edit]
Iminodiacetic acid is also used in the production of chelating resins, which are employed in the purification of metals and the treatment of wastewater. These resins are capable of selectively binding metal ions, facilitating their removal from complex mixtures.
Complexes with Metal Ions[edit]
IDA forms stable complexes with a variety of metal ions, including iron, copper, and zinc. These complexes are often used in analytical chemistry to determine the concentration of metal ions in solutions.
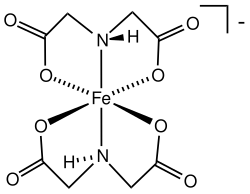
The ferric(IDA)2 anion is a notable example of such a complex, where the IDA ligands coordinate with a central iron ion. This complex is used in various analytical applications due to its stability and solubility in aqueous solutions.
Synthesis[edit]
Iminodiacetic acid can be synthesized through the reaction of chloroacetic acid with ammonia, followed by hydrolysis. This process involves the formation of an intermediate iminoacetic acid, which is then converted to the final product.
Related Compounds[edit]
IDA is related to other amino acids and chelating agents, such as ethylenediaminetetraacetic acid (EDTA) and nitrilotriacetic acid (NTA). These compounds share similar properties and applications, particularly in their ability to form stable complexes with metal ions.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
