Mepitiostane
Synthetic anabolic-androgenic steroid
| Mepitiostane | |
|---|---|
|
| |
| Chemical nomenclature | |
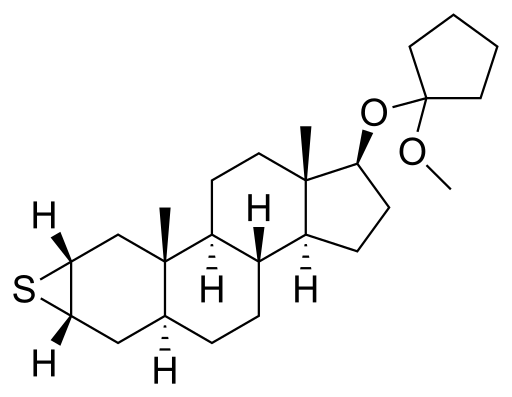
| IUPAC name | (2S,3aS,3bR,9aR,9bS,11aS)-2,3a,6,9,9-pentamethyl-2,3,3a,3b,4,5,6,7,8,9,9a,9b,10,11,11a-tetradecahydro-1H-cyclopenta[a]phenanthrene-2,7-diol
|
Mepitiostane is a synthetic anabolic-androgenic steroid (AAS) and a derivative of dihydrotestosterone (DHT). It is known for its use in the treatment of breast cancer in women. Mepitiostane is the 17α-methylated derivative of epitiostanol, which is also an AAS used for similar purposes.
Chemical Structure and Properties[edit]
Mepitiostane is chemically characterized by the presence of a 17α-methyl group, which distinguishes it from its parent compound, epitiostanol. This modification enhances its oral bioavailability, making it more effective when administered orally compared to non-17α-alkylated steroids.
The compound is a 17α-alkylated anabolic steroid, which means it has been modified to resist breakdown by the liver, allowing it to be effective when taken orally. However, this modification also increases the potential for hepatotoxicity, a common concern with 17α-alkylated steroids.
Pharmacology[edit]
Mepitiostane functions primarily as an androgen receptor agonist. It binds to androgen receptors in target tissues, promoting anabolic effects such as increased protein synthesis and muscle growth. Its androgenic effects are relatively mild compared to its anabolic effects, which makes it suitable for use in women, particularly in the treatment of breast cancer.
Medical Uses[edit]
Mepitiostane has been used in the treatment of breast cancer due to its ability to antagonize the effects of estrogen, a hormone that can promote the growth of certain types of breast cancer cells. By binding to androgen receptors, mepitiostane can inhibit the proliferation of these cancer cells.
Side Effects[edit]
As with other anabolic steroids, mepitiostane can cause a range of side effects. These may include virilization in women, such as deepening of the voice, increased body hair, and other masculinizing effects. Due to its 17α-alkylation, there is also a risk of liver damage with prolonged use.
Related Compounds[edit]
Mepitiostane is closely related to epitiostanol, which is its non-17α-alkylated counterpart. Both compounds share similar mechanisms of action and therapeutic uses, but mepitiostane's oral bioavailability makes it more convenient for certain treatment regimens.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian