Myopathic carnitine deficiency
Other names: carnitine transporter deficiency,carnitine uptake defect,carnitine uptake deficiency,CUD,renal carnitine transport defect,systemic carnitine deficiency
Primary carnitine deficiency is a condition that prevents the body from using certain fats for energy, particularly during periods without food (fasting). Carnitine, a natural substance acquired mostly through the diet, is used by cells to process fats and produce energy. Problems related to primary carnitine deficiency can be triggered by periods of fasting or by illnesses such as viral infections. This disorder is sometimes mistaken for Reye syndrome, a severe disorder that may develop in children while they appear to be recovering from viral infections such as chicken pox or flu. Most cases of Reye syndrome are associated with the use of aspirin during these viral infections.
Cause[edit]
Mutations in the SLC22A5 gene cause primary carnitine deficiency. This gene provides instructions for making a protein called OCTN2 that transports carnitine into cells. Cells need carnitine to bring certain types of fats (fatty acids) into mitochondria, which are the energy-producing centers within cells. Fatty acids are a major source of energy for the heart and muscles. During periods of fasting, fatty acids are also an important energy source for the liver and other tissues.
Mutations in the SLC22A5 gene result in an absent or dysfunctional OCTN2 protein. As a result, there is a shortage (deficiency) of carnitine within cells. Without carnitine, fatty acids cannot enter mitochondria and be used to make energy. Reduced energy production can lead to some of the features of primary carnitine deficiency, such as muscle weakness and hypoglycemia. Fatty acids may also build up in cells and damage the liver, heart, and muscles. This abnormal buildup causes the other signs and symptoms of the disorder.
Inheritance[edit]
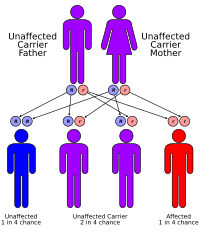
Primary carnitine deficiency is inherited in an autosomal recessive pattern, which means both copies of the gene in each cell have mutations. Most often, the parents of an individual with an autosomal recessive disorder are carriers, which means they each carry one copy of the mutated gene. Carriers of SLC22A5 gene mutations may have some signs and symptoms related to the condition.
Signs and symptoms[edit]
Signs and symptoms of primary carnitine deficiency typically appear during infancy or early childhood and can include severe brain dysfunction (encephalopathy), a weakened and enlarged heart (cardiomyopathy), confusion, vomiting, muscle weakness, and low blood sugar (hypoglycemia). The severity of this condition varies among affected individuals. Some people with primary carnitine deficiency are asymptomatic, which means they do not have any signs or symptoms of the condition. All individuals with this disorder are at risk for heart failure, liver problems, coma, and sudden death.
Diagnosis[edit]
The first suspicion of SPCD in a patient with a non-specific presentation is an extremely low plasma carnitine level. When combined with an increased concentration of carnitine in urine, the suspicion of SPCD can often be confirmed by either molecular testing or functional studies assessing the uptake of carnitine in cultured fibroblasts.
Treatment[edit]
Identification of patients presymptomatically via newborn screening has allowed early intervention and treatment. Treatment for SPCD involves high dose carnitine supplementation, which must be continued for life.Individuals who are identified and treated at birth have very good outcomes, including the prevention of cardiomyopathy. Mothers who are identified after a positive newborn screen but are otherwise asymptomatic are typically offered carnitine supplementation as well. The long-term outcomes for asymptomatic adults with SPCD is not known, but the discovery of mothers with undiagnosed cardiomyopathy and SPCD has raised the possibility that identification and treatment may prevent adult-onset manifestations. The medication(s) listed below have been approved by the Food and Drug Administration (FDA) as orphan products for treatment of this condition.
- Levocarnitine (Brand name: Carnitor®) Treatment of primary and secondary carnitine deficiency.
| Inborn error of lipid metabolism: fatty-acid metabolism disorders | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian