Pentamine
Pentamine[edit]
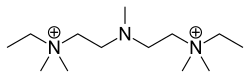
Pentamine is a chemical compound that belongs to the class of amines. It is characterized by the presence of five amine groups, which are nitrogen atoms bonded to hydrogen atoms or organic groups. Pentamine is of interest in various fields of chemistry and pharmacology due to its unique properties and potential applications.
Chemical Structure[edit]
Pentamine has a complex structure that includes five amine groups. The general formula for pentamines can be represented as R-NH_, where R is an organic group. The presence of multiple amine groups makes pentamines highly reactive and capable of forming multiple bonds with other chemical species.
Synthesis[edit]
The synthesis of pentamine compounds typically involves the reaction of primary amines with other chemical reagents that introduce additional amine groups. This process can be carried out through various methods, including alkylation, amination, and reductive amination.
Applications[edit]
Pentamines have a range of applications in different fields:
- Pharmaceuticals: In pharmacology, pentamines are studied for their potential use as drugs due to their ability to interact with biological molecules.
- Industrial Chemistry: They are used in the synthesis of polymers and other complex chemical structures.
- Research: Pentamines serve as important intermediates in the study of organic chemistry and biochemistry.
Biological Activity[edit]
Pentamines can exhibit significant biological activity due to their ability to interact with proteins, enzymes, and nucleic acids. This makes them potential candidates for drug development and other therapeutic applications.
Safety and Handling[edit]
As with many chemical compounds, proper safety measures should be taken when handling pentamines. They can be hazardous if not managed correctly, and appropriate personal protective equipment (PPE) should be used.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian