Preclinical development
Preclinical Development[edit]
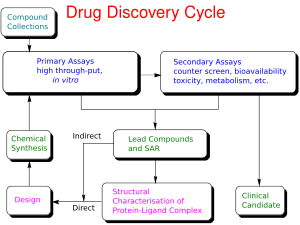
Preclinical development is a critical phase in the drug discovery process, where potential therapeutic compounds are tested extensively before they can proceed to clinical trials. This stage involves a series of laboratory and animal studies designed to assess the safety, efficacy, and pharmacokinetics of a drug candidate.
Objectives[edit]
The primary objectives of preclinical development are to:
- Determine the pharmacodynamics and pharmacokinetics of the drug candidate.
- Assess the toxicology and safety profile in animal models.
- Establish the formulation and stability of the drug.
- Gather data to support the design of clinical trials.
Key Components[edit]
Pharmacokinetics and Pharmacodynamics[edit]
Pharmacokinetics (PK) studies focus on how the drug is absorbed, distributed, metabolized, and excreted in the body. Pharmacodynamics (PD) studies, on the other hand, examine the biological effects of the drug and its mechanism of action. Together, PK/PD studies help in understanding the drug's behavior in biological systems.
Toxicology[edit]
Toxicology studies are conducted to identify any potential adverse effects of the drug candidate. These studies are crucial for determining the maximum tolerated dose and identifying any organ toxicity. They typically involve both acute and chronic exposure assessments in multiple animal species.
Formulation Development[edit]
Formulation development involves creating a stable and effective form of the drug that can be administered to patients. This includes determining the appropriate dosage form, such as tablets, capsules, or injectables, and ensuring the drug's stability over time.
Good Laboratory Practice (GLP)[edit]
Preclinical studies must adhere to Good Laboratory Practice (GLP) standards, which ensure the quality and integrity of the data collected. GLP compliance is essential for regulatory submissions and approval.
Regulatory Considerations[edit]
Before a drug candidate can move to clinical trials, a comprehensive Investigational New Drug (IND) application must be submitted to regulatory authorities, such as the Food and Drug Administration (FDA) in the United States. The IND application includes all preclinical data, proposed clinical trial protocols, and information on the drug's manufacturing process.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
