Triacetin
Triacetin[edit]
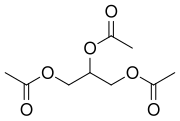
Triacetin, also known as glycerol triacetate, is a chemical compound that belongs to the class of compounds known as triesters. It is formed by the esterification of glycerol with three acetic acid molecules. Triacetin is a colorless, viscous liquid with a mild odor and is used in various industrial applications.
Chemical Properties[edit]
Triacetin has the chemical formula C9H14O6 and a molecular weight of 218.21 g/mol. It is soluble in water, alcohol, and ether. The compound has a boiling point of approximately 258 °C and a melting point of -78 °C.
Uses[edit]
Triacetin is used in a variety of applications due to its plasticizing, solvent, and fixative properties. Some of the primary uses include:
- As a plasticizer in the production of plastics and resins.
- As a solvent for cellulose acetate in the manufacture of cigarette filters.
- As a fixative in perfumery to stabilize fragrances.
- In the food industry as a food additive (E1518) to improve the texture and stability of baked goods and confectionery.
Biological Role[edit]
Triacetin is metabolized in the human body to glycerol and acetic acid, both of which are naturally occurring substances. It is considered non-toxic and is generally recognized as safe (GRAS) for use in food products.
Safety and Handling[edit]
While triacetin is considered safe for use in food and cosmetic products, it should be handled with care in its pure form. Appropriate safety measures should be taken to avoid inhalation or skin contact, and it should be stored in a cool, dry place away from incompatible substances.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian