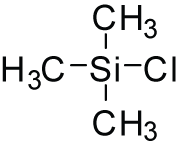Trimethylsilyl chloride
Trimethylsilyl Chloride[edit | edit source]
Trimethylsilyl chloride (TMSCl) is an organosilicon compound with the formula (CH_)_SiCl. It is a colorless liquid that is widely used in organic synthesis and as a reagent in the protection of functional groups.
Structure and Properties[edit | edit source]
Trimethylsilyl chloride is a silicon compound where the silicon atom is bonded to three methyl groups and one chlorine atom. The compound is volatile and has a boiling point of approximately 57 °C. It is soluble in organic solvents such as benzene, toluene, and diethyl ether.
Synthesis[edit | edit source]
TMSCl is typically produced by the reaction of silicon tetrachloride with methyl lithium or methyl magnesium chloride:
<math>\text{SiCl}_4 + 3 \text{CH}_3\text{MgCl} \rightarrow \text{(CH}_3\text{)}_3\text{SiCl} + 3 \text{MgCl}_2</math>
This reaction involves the substitution of three chlorine atoms in silicon tetrachloride with methyl groups.
Applications[edit | edit source]
Protection of Functional Groups[edit | edit source]
One of the primary uses of trimethylsilyl chloride is in the protection of hydroxyl groups in organic synthesis. It reacts with alcohols to form trimethylsilyl ethers, which are stable and can be deprotected under mild conditions:
<math>\text{ROH} + \text{(CH}_3\text{)}_3\text{SiCl} \rightarrow \text{ROSi(CH}_3\text{)}_3 + \text{HCl}</math>
Silylation Reagent[edit | edit source]
TMSCl is also used as a silylation reagent in the preparation of silyl enol ethers and other silylated compounds. These derivatives are useful intermediates in various chemical transformations.
Other Uses[edit | edit source]
In addition to its role in organic synthesis, trimethylsilyl chloride is used in the production of silicones and as a precursor to other organosilicon compounds.
Safety and Handling[edit | edit source]
Trimethylsilyl chloride is a corrosive substance and should be handled with care. It reacts with water to produce hydrochloric acid, which is corrosive to skin and eyes. Proper personal protective equipment (PPE) such as gloves and goggles should be used when handling this chemical.
Related Pages[edit | edit source]
Search WikiMD
Ad.Tired of being Overweight? Try W8MD's NYC physician weight loss.
Semaglutide (Ozempic / Wegovy and Tirzepatide (Mounjaro / Zepbound) available. Call 718 946 5500.
Advertise on WikiMD
|
WikiMD's Wellness Encyclopedia |
| Let Food Be Thy Medicine Medicine Thy Food - Hippocrates |
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
Medical Disclaimer: WikiMD is not a substitute for professional medical advice. The information on WikiMD is provided as an information resource only, may be incorrect, outdated or misleading, and is not to be used or relied on for any diagnostic or treatment purposes. Please consult your health care provider before making any healthcare decisions or for guidance about a specific medical condition. WikiMD expressly disclaims responsibility, and shall have no liability, for any damages, loss, injury, or liability whatsoever suffered as a result of your reliance on the information contained in this site. By visiting this site you agree to the foregoing terms and conditions, which may from time to time be changed or supplemented by WikiMD. If you do not agree to the foregoing terms and conditions, you should not enter or use this site. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Contributors: Prab R. Tumpati, MD