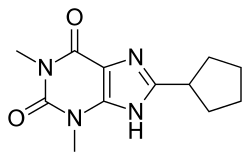
8-Cyclopentyl-1,3-dimethylxanthine
Chemical compound
| Chemical Compound | |
|---|---|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider ID | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| Properties | |
| Chemical Formula | |
| Molar Mass | |
| Appearance | |
| Density | |
| Melting Point | |
| Boiling Point | |
| Hazards | |
| GHS Pictograms | [[File:|50px]] |
| GHS Signal Word | |
| GHS Hazard Statements | |
| NFPA 704 | [[File:|50px]] |
| References | |
8-Cyclopentyl-1,3-dimethylxanthine is a chemical compound that belongs to the class of xanthine derivatives. It is known for its role as a potent and selective adenosine receptor antagonist, particularly targeting the A1 receptor subtype.
Chemical Structure and Properties[edit]
8-Cyclopentyl-1,3-dimethylxanthine has a molecular structure that includes a xanthine core with a cyclopentyl group at the 8-position and two methyl groups at the 1- and 3-positions. The chemical formula is C13H18N4O2.
Mechanism of Action[edit]
As an adenosine receptor antagonist, 8-Cyclopentyl-1,3-dimethylxanthine inhibits the action of adenosine at the A1 receptor. This inhibition can lead to various physiological effects, including increased neurotransmitter release and enhanced neuronal activity.
Pharmacological Effects[edit]
The compound is primarily used in research to study the role of adenosine in the central nervous system and its effects on neurotransmission. It has been shown to have potential applications in the treatment of neurological disorders and cardiovascular diseases.
Research Applications[edit]
8-Cyclopentyl-1,3-dimethylxanthine is widely used in pharmacological studies to investigate the function of adenosine receptors. It serves as a tool to understand the physiological and pathological roles of adenosine in various tissues.
Safety and Toxicity[edit]
The safety profile of 8-Cyclopentyl-1,3-dimethylxanthine is not well-documented, and it should be handled with care in a laboratory setting. Proper safety protocols should be followed to avoid any potential hazards.
See Also[edit]
References[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian