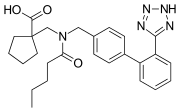
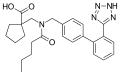
Abitesartan
A detailed overview of the pharmaceutical drug Abitesartan
| Abitesartan | |
|---|---|
|
|
Abitesartan is a pharmaceutical drug that belongs to the class of angiotensin II receptor antagonists, commonly known as ARBs. It is primarily used in the management of hypertension and certain types of heart failure.
Mechanism of Action[edit]
Abitesartan works by selectively blocking the binding of angiotensin II to the angiotensin II receptor type 1 (AT1 receptor) in various tissues, including the vascular smooth muscle, adrenal gland, and kidney. This inhibition prevents the vasoconstrictor and aldosterone-secreting effects of angiotensin II, leading to a decrease in blood pressure.
Pharmacokinetics[edit]
Abitesartan is administered orally and is well absorbed from the gastrointestinal tract. It undergoes minimal first-pass metabolism and is primarily excreted unchanged in the urine and feces. The drug has a half-life of approximately 12 hours, allowing for once-daily dosing.
Clinical Uses[edit]
Hypertension[edit]
Abitesartan is indicated for the treatment of hypertension to lower blood pressure and reduce the risk of cardiovascular events such as stroke and myocardial infarction.
Heart Failure[edit]
In patients with heart failure, Abitesartan is used to improve symptoms and reduce the risk of hospitalization and mortality.
Side Effects[edit]
Common side effects of Abitesartan include dizziness, headache, and fatigue. Rare but serious side effects may include angioedema and renal impairment.
Contraindications[edit]
Abitesartan is contraindicated in patients with known hypersensitivity to the drug or any of its components. It should not be used during pregnancy due to the risk of fetal harm.
Related pages[edit]
-
Abitesartan
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian