Bolasterone
Anabolic steroid
Bolasterone[edit]
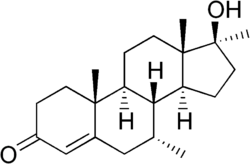
Bolasterone is a synthetic anabolic steroid that was first developed in the 1950s. It is known for its ability to promote muscle growth and increase strength, making it popular among bodybuilders and athletes. However, its use is associated with significant side effects and it is classified as a controlled substance in many countries.
Chemical Properties[edit]
Bolasterone is a derivative of testosterone, the primary male sex hormone and anabolic steroid. It is chemically known as 7_,17_-dimethyl-17_-hydroxyandrost-4-en-3-one. The addition of methyl groups at the 7_ and 17_ positions increases its oral bioavailability and anabolic activity.
Pharmacology[edit]
Bolasterone exhibits strong anabolic effects with moderate androgenic properties. It binds to the androgen receptors in muscle tissue, promoting protein synthesis and muscle growth. Its anabolic to androgenic ratio is higher than that of testosterone, making it more effective in building muscle mass with fewer androgenic side effects.
Medical Uses[edit]
Historically, bolasterone was used in the treatment of conditions such as muscle wasting and osteoporosis. However, due to its potential for abuse and adverse effects, its medical use has become limited.
Side Effects[edit]
The use of bolasterone can lead to a range of side effects, including:
- Liver toxicity due to its 17_-alkylated structure.
- Gynecomastia (development of breast tissue in men) due to its estrogenic activity.
- Acne and oily skin as a result of increased sebum production.
- Hair loss and hirsutism (excessive hair growth) due to its androgenic effects.
- Suppression of natural testosterone production, leading to hypogonadism.
Legal Status[edit]
Bolasterone is classified as a Schedule III controlled substance under the Anabolic Steroid Control Act in the United States. It is banned by most major sports organizations, including the World Anti-Doping Agency (WADA).
Related Compounds[edit]
Bolasterone is structurally related to other anabolic steroids such as methandrostenolone and oxandrolone. These compounds share similar anabolic properties but differ in their androgenic effects and side effect profiles.
See Also[edit]
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian