Glyceraldehyde
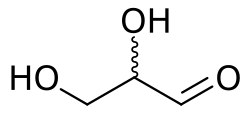
Glyceraldehyde is a simple sugar and an important intermediate in several metabolic pathways. It is a triose, meaning it contains three carbon atoms. The chemical formula for glyceraldehyde is C₃H₆O₃. It exists in two enantiomers, D-glyceraldehyde and L-glyceraldehyde, which are mirror images of each other.
Structure and Stereochemistry[edit]
Glyceraldehyde is the simplest of all aldoses, which are sugars containing an aldehyde group. The molecule has a single chiral center at the second carbon atom, leading to two possible stereoisomers. The D- and L- forms of glyceraldehyde are named based on their relationship to the D- and L- nomenclature system used for carbohydrates.
Biological Role[edit]
Glyceraldehyde plays a crucial role in the glycolysis pathway, where it is involved in the breakdown of glucose to produce energy. It is also a key intermediate in the Calvin cycle of photosynthesis and in the pentose phosphate pathway.
Metabolic Pathways[edit]
In glycolysis, glyceraldehyde is formed from the cleavage of fructose-1,6-bisphosphate into two three-carbon molecules: dihydroxyacetone phosphate (DHAP) and glyceraldehyde-3-phosphate (G3P). G3P is then further metabolized to produce pyruvate, which enters the citric acid cycle.
In the Calvin cycle, glyceraldehyde-3-phosphate is one of the products of the reduction phase, where it is used to regenerate ribulose-1,5-bisphosphate and to synthesize glucose and other carbohydrates.
Chemical Properties[edit]
Glyceraldehyde is a highly reactive molecule due to its aldehyde group. It can undergo various chemical reactions, including oxidation to form glyceric acid and reduction to form glycerol. It can also participate in aldol reactions and other carbon-carbon bond-forming reactions.
Applications[edit]
Glyceraldehyde is used in biochemical research to study enzyme kinetics and metabolic pathways. It is also used as a standard in chromatography and other analytical techniques.
See Also[edit]
References[edit]
External Links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian