Lecanemab
What is Lecanemab?[edit]
- Lecanemab (Leqembi) is a recombinant humanized immunoglobulin gamma 1 (IgG1) monoclonal antibody directed against aggregated soluble and insoluble forms of amyloid beta, and is expressed in a Chinese hamster ovary cell line.
What are the uses of this medicine?[edit]
Lecanemab (Leqembi) is a prescription medicine used to treat people with Alzheimer’s disease.
Limitations of use:
- Treatment with Leqembi should be initiated in patients with mild cognitive impairment or mild dementia stage of disease.
- There are no safety or effectiveness data on initiating treatment at earlier or later stages of the disease than were studied.
How does this medicine work?[edit]
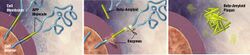
- Lecanemab-irmb is a humanized immunoglobulin gamma 1 (IgG1) monoclonal antibody directed against aggregated soluble and insoluble forms of amyloid beta.
- The accumulation of amyloid beta plaques in the brain is a defining pathophysiological feature of Alzheimer’s disease.
- Leqembi reduces amyloid beta plaques.
Who Should Not Use this medicine ?[edit]
- This medicine have no usage limitations.
What drug interactions can this medicine cause?[edit]
- Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Is this medicine FDA approved?[edit]
- Lecanemab was approved for medical use in the United States in January 2023.
How should this medicine be used?[edit]
- Confirm the presence of amyloid beta pathology prior to initiating treatment.
- Obtain a recent (within one year) brain MRI prior to initiating treatment to evaluate for pre-existing Amyloid Related Imaging Abnormalities (ARIA).
Recommended dosage:
- The recommended dosage of Leqembi is 10 mg/kg that must be diluted then administered as an intravenous infusion over approximately one hour, once every two weeks.
- Obtain an MRI prior to the 5th, 7th, and 14th infusions.
- If radiographically observed ARIA occurs, treatment recommendations are based on type, severity, and presence of symptoms.
Administration:
- Leqembi is given by a healthcare provider through a needle placed in your vein (intravenous (IV) infusion) in your arm.
- Leqembi is given every 2 weeks. Each infusion will last about 1 hour.
- If you miss an infusion of Leqembi, you should receive your next dose as soon as possible.
What are the dosage forms and brand names of this medicine?[edit]
This medicine is available in fallowing doasage form: As Injection:
- 500 mg/5 mL (100 mg/mL) solution in a single-dose vial
- 200 mg/2 mL (100 mg/mL) solution in a single-dose vial
This medicine is available in fallowing brand namesː
- Leqembi
What side effects can this medication cause?[edit]
The most common side effects of this medicine include:
- infusion-related reactions
- headache
- swelling in areas of the brain, with or without small spots of bleeding in or on the surface of the brain (ARIA)
Less common, but serious side effects may include:
- Amyloid Related Imaging Abnormalities or “ARIA”
- infusion-related reactions
What special precautions should I follow?[edit]
- Monoclonal antibodies directed against aggregated forms of beta amyloid, including Leqembi, can cause amyloid related imaging abnormalities (ARIA). Inform patients that Leqembi may cause Amyloid Related Imaging Abnormalities or “ARIA”.
- Advise patients of the potential risk of infusion-related reactions, which can include flu-like symptoms, nausea, vomiting, and changes in blood pressure, the majority of which occur with the first infusion.
- Advise patients that the Alzheimer’s Network for Treatment and Diagnostics (ALZ-NET) is a voluntary provider-enrolled patient registry that collects information on treatments for Alzheimer’s disease, including Leqembi. Encourage patients to participate in the ALZ-NET registry.
- It is not known if lecanemab-irmb passes into your breast milk. Talk to your healthcare provider about the best way to feed your baby while receiving Leqembi.
What to do in case of emergency/overdose?[edit]
- In case of overdose, call the poison control helpline of your country. In the United States, call 1-800-222-1222.
- Overdose related information is also available online at poisonhelp.org/help.
- In the event that the victim has collapsed, had a seizure, has trouble breathing, or can't be awakened, immediately call emergency services. In the United States, call 911.
- In the event of overdose, general supportive measures should be instituted as necessary.
- Monitor the patient for adverse reactions.
Can this medicine be used in pregnancy?[edit]
- It is not known if Leqembi will harm your unborn baby. Tell your healthcare provider if you become pregnant during your treatment with Leqembi.
Can this medicine be used in children?[edit]
- Safety and effectiveness of Leqembi in pediatric patients have not been established.
What are the active and inactive ingredients in this medicine?[edit]
- Active ingredient: lecanemab-irmb.
- Inactive ingredients: arginine hydrochloride, histidine, histidine hydrochloride monohydrate, polysorbate 80, and water for injection.
Who manufactures and distributes this medicine?[edit]
Manufactured by:
- Eisai Inc.
- Nutley, NJ
What should I know about storage and disposal of this medication?[edit]
Unopened Vial:
- Store in a refrigerator at 2°C to 8°C (36°F to 46°F).
- Store in the original carton to protect from light.
- Do not freeze or shake.
Diluted Solution:
- After dilution, immediate use is recommended.
- If not administered immediately, store Leqembi refrigerated at 2°C to 8°C (36°F to 46°F) for up to 4 hours, or at room temperature up to 30°C (86°F) for up to 4 hours.
- Do not freeze.
| Monoclonals for bone, musculoskeletal, circulatory, and neurologic systems | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian