Medifoxamine
An atypical antidepressant
| Medifoxamine | |
|---|---|
[[File: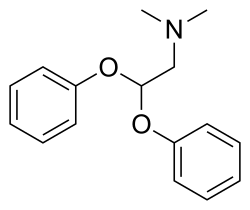
| |
| Trade names | Clédial, Gerdaxyl
|
| Chemical nomenclature | |
| IUPAC name | 2-(3,4-dimethoxyphenyl)-2-methylaminopropan-1-one
|
| Routes | Oral
|
| Metabolism | Hepatic
|
| Excretion | Renal |
| Legal status | Rx-only |
| Identifiers | |
| CAS Number | 3239-44-9 |
| PubChem | 4053 |
| DrugBank | DB08998 |
| ChemSpider | 3913 |
| UNII | 0T493YFU8O |
| KEGG | D07355 |
| ChEMBL | 2104660 |
| Chemical data
| |
| Chemical formula | C12H17N1O3
|
| SMILES | CC(C(=O)C1=CC(=C(C=C1)OC)OC)NC
|
| InChI | 1S/C12H17NO3/c1-8(13-2)12(14)9-5-6-10(15-3)11(7-9)16-4/h5-8,13H,1-4H3
|
| InChIKey | ZJQJHFOYQZKEMV-UHFFFAOYSA-N
|
Medifoxamine is an atypical antidepressant that was developed in the 1970s and used primarily in France and other European countries. It is known for its unique pharmacological profile, which distinguishes it from other antidepressants such as selective serotonin reuptake inhibitors (SSRIs) and tricyclic antidepressants (TCAs).
Pharmacology[edit]
Medifoxamine acts as a serotonin and dopamine reuptake inhibitor, which contributes to its antidepressant effects. Unlike many other antidepressants, it does not significantly affect the reuptake of norepinephrine. This selective action is thought to reduce the incidence of certain side effects commonly associated with other antidepressant classes.
Additionally, medifoxamine has been shown to possess anxiolytic properties, making it potentially useful in the treatment of anxiety disorders. Its mechanism of action also includes modulation of GABAergic activity, which may contribute to its calming effects.
Clinical Use[edit]
Medifoxamine was primarily prescribed for the treatment of major depressive disorder and anxiety disorders. It was favored for its relatively mild side effect profile compared to other antidepressants available at the time. Patients reported fewer issues with sedation, weight gain, and sexual dysfunction, which are common side effects of many antidepressants.
Side Effects[edit]
While medifoxamine is generally well-tolerated, some patients may experience side effects such as nausea, dizziness, and headache. Rarely, it may cause allergic reactions or hepatic issues, necessitating regular monitoring of liver function during treatment.
Discontinuation[edit]
Medifoxamine was eventually withdrawn from the market due to concerns over hepatotoxicity, which is the potential for causing liver damage. Despite its efficacy and favorable side effect profile, the risk of liver damage led to its discontinuation in the late 1990s.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian