Opiorphin
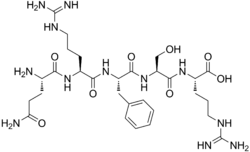
Opiorphin is a peptide that was first isolated from human saliva. It is a relatively small molecule, consisting of only five amino acids, but it has been found to have potent analgesic properties.
Discovery[edit]
Opiorphin was discovered in 2006 by a team of researchers led by Catherine Rougeot at the Institut Pasteur in Paris, France. The team was investigating the analgesic properties of human saliva when they isolated the peptide.
Structure and Function[edit]
Opiorphin is a pentapeptide, meaning it is composed of five amino acids. These are glutamine, arginine, proline, arginine, and proline (QRPRL). The peptide is synthesized in the salivary glands and secreted into the saliva.
The primary function of opiorphin is to inhibit the breakdown of enkephalins, which are endogenous opioid peptides. It does this by inhibiting two enzymes, neutral endopeptidase (NEP) and aminopeptidase N (APN), which are responsible for degrading enkephalins. By preventing the breakdown of these peptides, opiorphin increases their concentration in the body, leading to increased analgesic effects.
Potential Therapeutic Uses[edit]
Due to its analgesic properties, opiorphin has potential for use in the treatment of pain. In animal studies, it has been found to be as effective as morphine in relieving pain, but without the side effects associated with opioid drugs. However, further research is needed to determine its safety and efficacy in humans.
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian