Salt (chemistry)
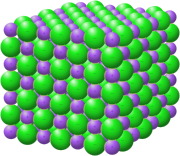

Salt in chemistry refers to an ionic compound that results from the neutralization reaction of an acid and a base. Salts are composed of cations (positively charged ions) and anions (negatively charged ions), which combine to form a neutral product.
Formation[edit]
Salts are typically formed through the reaction of an acid and a base. This process is known as neutralization. For example, when hydrochloric acid (HCl) reacts with sodium hydroxide (NaOH), the result is sodium chloride (NaCl) and water (H₂O):
- HCl + NaOH → NaCl + H₂O
Types of Salts[edit]
Salts can be classified into several types based on their properties and the nature of their constituent ions:
- Simple salts: Formed from the neutralization of a single acid and a single base. Example: sodium chloride (NaCl).
- Double salts: Contain more than one type of cation or anion. Example: alum (KAl(SO₄)₂·12H₂O).
- Complex salts: Contain complex ions. Example: potassium ferricyanide (K₃[Fe(CN)₆]).
- Acid salts: Formed by the partial neutralization of a diprotic or polyprotic acid. Example: sodium bisulfate (NaHSO₄).
- Basic salts: Formed by the partial neutralization of a polyacidic base. Example: bismuth subnitrate (BiONO₃).
Properties[edit]
Salts exhibit a wide range of properties:
- Solubility: Many salts are soluble in water, but some are not. For example, sodium chloride is highly soluble, while silver chloride (AgCl) is not.
- Electrical conductivity: In their molten state or when dissolved in water, salts conduct electricity due to the movement of ions.
- Melting point: Salts generally have high melting points due to the strong ionic bonds between the cations and anions.
Applications[edit]
Salts have numerous applications in various fields:
- Food industry: Sodium chloride is widely used as a seasoning and preservative.
- Chemical industry: Salts are used in the production of various chemicals. For example, sodium carbonate (Na₂CO₃) is used in glass manufacturing.
- Medicine: Certain salts, such as magnesium sulfate (Epsom salt), are used for their therapeutic properties.
See also[edit]
References[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
