Xylitol
A sugar alcohol used as a sweetener
Xylitol[edit]
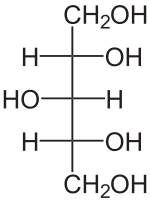
Xylitol is a sugar alcohol used as a sweetener. It is a colorless or white crystalline solid that is soluble in water. Xylitol is categorized as a polyol and is used as a sugar substitute in various products, including chewing gum, toothpaste, and mouthwash.
Chemical Structure and Properties[edit]
Xylitol has the chemical formula C5H12O5. It is an achiral compound, meaning it does not have a chiral center. The structure of xylitol consists of a five-carbon backbone with hydroxyl groups attached to each carbon, making it a pentitol.
Production[edit]
Xylitol is produced industrially by the hydrogenation of xylose, which is derived from hemicellulose. The process involves the catalytic reduction of xylose to xylitol, typically using a nickel catalyst.
Uses[edit]
Xylitol is widely used as a sugar substitute due to its sweetness, which is comparable to that of sucrose, but with fewer calories. It is commonly found in "sugar-free" products, such as:
Health Benefits[edit]
Xylitol is known for its dental benefits. It is non-cariogenic, meaning it does not contribute to tooth decay. In fact, xylitol can help reduce the levels of Streptococcus mutans, a bacterium associated with cavities, in the mouth. It also promotes the remineralization of tooth enamel.
Safety and Side Effects[edit]
Xylitol is generally considered safe for human consumption. However, excessive intake can lead to gastrointestinal discomfort, such as bloating and diarrhea, due to its osmotic effect in the intestine.
Toxicity in Animals[edit]
Xylitol is highly toxic to dogs and can cause a rapid release of insulin, leading to hypoglycemia and potentially liver failure. Pet owners should be cautious to keep xylitol-containing products away from their pets.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian