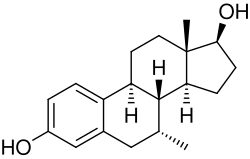
7α-Methylestradiol
Synthetic estrogen
| 7α-Methylestradiol
| |
|---|---|
| Synonyms | 7α-Methyl-17β-estradiol; 7α-Methyl-E2; 7α-Methylestra-1,3,5(10)-triene-3,17β-diol |
| Chemical nomenclature | |
| IUPAC name | (8R,9S,13S,14S,17S)-13-methyl-6,7,8,9,11,12,14,15,16,17-decahydrocyclopenta[a]phenanthrene-3,17-diol
|
| Identifiers | |
| CAS Number | 13110-72-4 |
| PubChem | 66435
|
| ChemSpider | 59808
|
| ChEMBL | 303926 |
| Chemical data
| |
| Chemical formula | C19H26O2
|
| SMILES | CC12CCC3C(C1CCC2O)CCC4=C3C=CC(=C4)O
|
7α-Methylestradiol is a synthetic estrogen and a derivative of estradiol. It is characterized by the presence of a methyl group at the 7α position of the estradiol molecule. This modification alters its pharmacokinetics and pharmacodynamics compared to estradiol.
Chemical Structure[edit]
7α-Methylestradiol is a steroid with the IUPAC name (8R,9S,13S,14S,17S)-13-methyl-6,7,8,9,11,12,14,15,16,17-decahydrocyclopenta[a]phenanthrene-3,17-diol. Its molecular formula is C19H26O2, and it has a molecular weight of 286.41 g/mol.
Pharmacology[edit]
As an estrogen, 7α-Methylestradiol binds to and activates the estrogen receptors (ERs), which are involved in the regulation of various physiological processes. The addition of the 7α-methyl group is intended to enhance its bioavailability and metabolic stability.
Uses[edit]
7α-Methylestradiol is primarily used in research settings to study the effects of estrogens and their receptors. It is not commonly used in clinical practice.
Synthesis[edit]
The synthesis of 7α-Methylestradiol involves the chemical modification of estradiol through the introduction of a methyl group at the 7α position. This process typically requires several steps, including protection and deprotection of functional groups, as well as oxidation and reduction reactions.
Related Compounds[edit]
See Also[edit]
References[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian