Aconitine
Aconitine
Aconitine is a highly toxic alkaloid derived from various species of the Aconitum plant, commonly known as monkshood or wolfsbane. It is known for its potent neurotoxic effects and has been historically used as a poison and in traditional medicine.
Chemical Structure[edit]
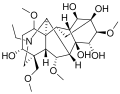
Aconitine is a member of the diterpene alkaloids, characterized by its complex polycyclic structure. The chemical formula of aconitine is C34H47NO11. It contains multiple ester groups and a nitrogen atom, which contribute to its biological activity.
Sources[edit]
Aconitine is primarily found in the roots and tubers of Aconitum species. These plants are native to mountainous regions of the Northern Hemisphere, including parts of Europe, Asia, and North America.
Mechanism of Action[edit]
Aconitine exerts its toxic effects by binding to the voltage-gated sodium channels in nerve cells. This binding causes persistent activation of the channels, leading to continuous depolarization of the nerve cells. This results in neurological symptoms such as paresthesia, muscle weakness, and potentially fatal arrhythmias.
Toxicity[edit]
Aconitine is extremely toxic, with a lethal dose in humans estimated to be around 2 mg. Symptoms of aconitine poisoning include nausea, vomiting, diarrhea, dizziness, and cardiac arrest. Due to its high toxicity, aconitine is not used in modern medicine but has been used historically in traditional Chinese medicine and as a poison.
Historical Use[edit]
Historically, aconitine has been used as a poison in various cultures. It was used on arrows for hunting and warfare. In traditional medicine, it was used in small doses for its analgesic and anti-inflammatory properties, although such use is highly risky due to its narrow therapeutic index.
Synthesis[edit]
The synthesis of aconitine is complex due to its intricate structure. The first total synthesis was achieved by Wiesner and his team, which involved multiple steps to construct the polycyclic framework and introduce the necessary functional groups.
Metabolism[edit]
In the body, aconitine is metabolized primarily in the liver. It undergoes hydrolysis to form less toxic metabolites, such as aconine. However, the rapid onset of its toxic effects often precedes significant metabolism.
Related Compounds[edit]
Aconitine is related to other alkaloids found in Aconitum species, such as mesaconitine and hypaconitine, which share similar toxicological profiles.
Related Pages[edit]
Gallery[edit]
-
Structure of Aconine, a metabolite of Aconitine
-
Aconitine 2D Structure
-
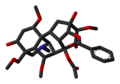
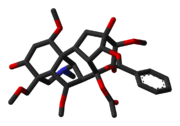
Aconitine Crystal 3D Sticks Skeletal
-
Wiesner Syntheses Schematic
-
Aconine
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian