Glycogen storage disease type II
| Glycogen storage disease type II | |
|---|---|
| Pompe vacuoles.jpg | |
| Muscle biopsy showing large vacuoles typical of Pompe disease (HE stain, frozen section). | |
| Synonyms | Pompe disease, acid maltase deficiency |
| Pronunciation | Pompe
|
| Symptoms | Progressive muscle weakness, cardiomegaly, respiratory difficulties |
| Complications | Heart failure, respiratory failure |
| Usual onset | Varies (infantile to adult onset) |
| Duration | Lifelong |
| Types | Infantile-onset, late-onset |
| Causes | Deficiency of lysosomal acid alpha-glucosidase enzyme |
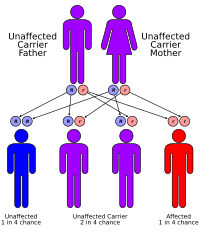
| Risk factors | Family history (autosomal recessive inheritance) |
| Diagnosis | Genetic testing, enzyme assay, muscle biopsy |
| Differential diagnosis | Other glycogen storage diseases, muscular dystrophies |
| Prevention | Genetic counseling |
| Treatment | Enzyme replacement therapy, supportive care
|
| Prognosis | Varies widely, severe forms have reduced life expectancy |
| Frequency | Rare |
| Deaths | Rare, dependent on disease severity and management |
Glycogen storage disease type II, also known as Pompe disease or acid maltase deficiency, is a rare, inherited metabolic disorder characterized by the accumulation of glycogen in body tissues due to a deficiency of the enzyme lysosomal acid alpha-glucosidase. This buildup results in progressive damage primarily to muscle and nerve tissues.
History[edit]
Pompe disease was first described in 1932 by the Dutch pathologist J.C. Pompe, marking it as the first identified glycogen storage disease. Pompe observed glycogen accumulation within muscle tissue in affected infants.
Classification[edit]
Pompe disease is classified into two main types:
- Infantile-onset Pompe disease: Severe, rapidly progressive form presenting within the first few months of life with marked cardiomegaly, muscle weakness, and respiratory issues.
- Late-onset Pompe disease: Presents in childhood, adolescence, or adulthood with a more gradual progression primarily affecting skeletal muscles and respiratory function.
Clinical Features[edit]
Common symptoms include:
- Progressive muscle weakness (myopathy)
- Respiratory difficulties
- Enlarged heart (cardiomegaly) primarily in infantile-onset
- Impaired motor skills and mobility
Pathophysiology[edit]
The deficiency of acid alpha-glucosidase enzyme disrupts lysosomal glycogen breakdown, causing excessive glycogen storage. Over time, this results in cellular dysfunction and muscle fiber damage, impacting skeletal, cardiac, and smooth muscle tissues.
Diagnosis[edit]
Diagnostic approaches include:
- Genetic testing to detect mutations in the GAA gene
- Enzyme assays to measure acid alpha-glucosidase activity
- Muscle biopsy showing glycogen accumulation and characteristic vacuoles
Treatment[edit]
Current treatment strategies include:
- Enzyme replacement therapy (ERT), specifically recombinant acid alpha-glucosidase, which can slow disease progression.
- Supportive therapies, including physical therapy, respiratory support, nutritional management, and cardiac care.
Prognosis[edit]
Prognosis varies significantly depending on the age of onset, severity, and timely initiation of treatment. Infantile-onset disease typically has a poor prognosis without early intervention, while individuals with late-onset forms may have a near-normal lifespan with appropriate management.
Prevention[edit]
Given the autosomal recessive inheritance, genetic counseling is recommended for families with a history of Pompe disease to evaluate risks for future offspring.
See Also[edit]
- Glycogen storage diseases
- Lysosomal storage disorders
- Metabolic disorders
- Enzyme replacement therapy
| Inborn error of carbohydrate metabolism: monosaccharide metabolism disorders (E73–E74, 271) Including glycogen storage diseases (GSD) |
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian