2-Butanol
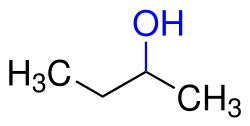
2-Butanol (also known as sec-butanol) is an organic compound with the formula CH3CH(OH)CH2CH3. This secondary alcohol is a flammable, colorless liquid that is soluble in 12 parts water and completely miscible with polar organic solvent such as ethers and other alcohols. It is produced on a large scale, primarily as a precursor to the industrial solvent methyl ethyl ketone. 2-Butanol is chiral and thus can be obtained as either of two stereoisomers.
Production[edit]
2-Butanol is produced by the hydration of butene in a process that involves acid catalysts. The reaction is highly exothermic, with a heat of reaction of -216 kJ/mol. The process is typically carried out in the liquid phase at elevated pressure.
Uses[edit]
2-Butanol is used as a solvent for many chemical reactions, as well as being a precursor to other chemical compounds. It is also used in the manufacture of brake fluid and as a component of perfumes due to its pleasant odor.
Safety[edit]
2-Butanol is considered a mild skin irritant and may cause eye irritation. It is also considered a mild central nervous system depressant and can cause dizziness, headache, and nausea if inhaled in high concentrations.
See also[edit]
References[edit]
| Chemical compounds | ||||||
|---|---|---|---|---|---|---|
This chemical compound-related article is a stub.
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
