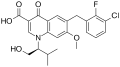
Elvitegravir
Information about Elvitegravir[edit]
Elvitegravir is a human immunodeficiency virus (HIV) integrase inhibitor which is used largely in a four drug combination with cobicistat, emtricitabine and tenofovir as therapy of HIV infection.
Liver safety of Elvitegravir[edit]
Therapy with this elvitegravir based regimen is often associated with transient serum aminotransferase elevations during therapy, but has not been implicated in cases of clinically apparent acute liver injury.
Mechanism of action of Elvitegravir[edit]
Elvitegravir (el" vi teg' ra vir) is a 4-quinolone-3-glyoxylic acid and antiretroviral agent that acts by inhibition of viral DNA strand transfer by the HIV integrase, a necessary step in HIV replication. Elvitegravir has been shown to lower serum levels of HIV RNA and to raise CD4 counts. In multiple prospective clinical trials, the combination of elvitegravir with tenofovir and emtricitabine has been found to be as effective as other standard antiretroviral combinations. Elvitegravir is given with cobicistat, a pharmacokinetic enhancer that inhibits CYP 3A4 activity, causing increased levels and more prolonged activity of drugs like elvitegravir that are metabolized by the hepatic cytochrome P450 isomer CYP 3A4.
FDA approval information for Elvitegravir[edit]
Elvitegravir was approved as a part of a four drug combination including cobicistat, emtricitabine and tenofovir disoproxil fumarate as therapy of HIV infection in 2012 under the brand name Stribild. A similar four drug combination that includes tenofovir alafenamide instead of tenofovir disoproxil fumarate was approved for use in HIV infection in 2016 under the brand name Genvoya. These combinations are available as tablets of 150 mg of elvitegravir with 150 mg of cobicistat, 200 mg of emtricitabine and either 300 mg of tenofovir disoproxil fumarate (Stribild) or 10 mg of tenofovir alafenamide (Genvoya). The recommended dose is one tablet daily, these combination being two of several "single tablet regimens" (STRs) for therapy of HIV infection. Elvitegravir was the second HIV integrase to be approved in the United States and shares structural similarity and resistance patterns with the initial agent, raltegravir. Elvitegravir is also available as a single agent for use with other antiretroviral agents in 85 and 150 mg tablets under the brand name Vitekta.
Side effects of Elvitegravir[edit]
Common side effects of the four drug combination include fatigue, diarrhea, nausea, dizziness, headache, depression, abnormal dreams and skin rashes. Cobicistat has inhibitory activity against several drug metabolizing enzymes besides CYP 3A4, including CYP 2D6 and the P-glycoprotein transporter, making it likely to cause drug-drug interactions and important to avoid when using other agents that are metabolized by the P450 system. Cobicistat also inhibits creatinine secretion, which artificially raises serum creatinine levels without affecting the glomerular filtration rate. With long term use, tenofovir disoproxil fumarate can be associated with decline in kidney function, phosphate wasting and decline in bone mineral density. These adverse effects appear to be less with tenofovir alafenamide which is more potent and is given in a lower dose that the disoproxil fumurate form of tenofovir.
Antiviral agents[edit]
Drugs for HIV Infection, in the Subclass Antiretroviral Agents
- Fusion Inhibitors (HIV)
- Integrase Inhibitors (HIV)
- Nonnucleoside Reverse Transcriptase Inhibitors (HIV)
- Interferon Based Therapies
HCV NS5A Inhibitors
HCV NS5B (Polymerase) Inhibitors
- Asunaprevir, Boceprevir, Glecaprevir, Grazoprevir, Paritaprevir, Simeprevir, Telaprevir, Voxilaprevir
Combination Therapies
Drugs for Herpes Virus Infections (HSV, CMV, others)
Drugs for Influenza
-
Structure of Elvitegravir
-
3D model of Elvitegravir
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian