Molnupiravir
Molnupiravir[edit]
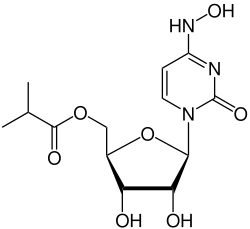
Molnupiravir is an antiviral medication that is used to treat COVID-19. It was developed by the pharmaceutical companies Merck & Co. and Ridgeback Biotherapeutics. Molnupiravir is a prodrug of the synthetic nucleoside derivative N4-hydroxycytidine and exerts its antiviral action by introducing errors into the viral RNA during replication.
Mechanism of Action[edit]
Molnupiravir works by incorporating itself into the viral RNA. Once inside the viral RNA, it causes mutations during the replication process. These mutations accumulate and lead to "error catastrophe," which reduces the ability of the virus to replicate and spread.
The drug is metabolized into its active form, N4-hydroxycytidine, which is then phosphorylated to the active triphosphate form. This active form is incorporated into the viral RNA by the viral RNA-dependent RNA polymerase.
Chemical Properties[edit]
Molnupiravir is a prodrug, meaning it is metabolized in the body to become an active pharmacological agent. Its chemical structure allows it to mimic the natural nucleosides used by the virus to replicate its RNA.
Synthesis[edit]
The synthesis of molnupiravir involves several chemical reactions starting from uridine, a naturally occurring nucleoside. The process includes the introduction of a hydroxylamine group to form N4-hydroxycytidine, which is then converted into molnupiravir.
Bonding and Interaction with Viral RNA[edit]
Molnupiravir can pair with both guanine and adenine, leading to G-to-A and C-to-U transitions in the viral genome. This dual pairing ability is crucial for its mechanism of inducing lethal mutagenesis in the virus.
Clinical Use[edit]
Molnupiravir is administered orally and is used in the treatment of mild to moderate COVID-19 in adults who are at risk for progressing to severe disease. It is typically prescribed for patients who cannot receive other treatments or for whom other treatments are not appropriate.
Safety and Efficacy[edit]
Clinical trials have shown that molnupiravir can reduce the risk of hospitalization and death in patients with COVID-19. However, its use is subject to ongoing evaluation, and it is important to consider potential side effects and contraindications.
Related Pages[edit]
Drugs for HIV Infection, in the Subclass Antiretroviral Agents
- Fusion Inhibitors (HIV)
- Integrase Inhibitors (HIV)
- Nonnucleoside Reverse Transcriptase Inhibitors (HIV)
- Nucleoside Analogues (HIV)
- Protease Inhibitors (HIV)
HCV NS5A Inhibitors
HCV NS5B Inhibitors (Polymerase inhibitors)
- Asunaprevir, Boceprevir, Glecaprevir, Grazoprevir, Paritaprevir, Simeprevir, Telaprevir, Voxilaprevir
Combination Therapies
Drugs for Herpes Virus
- infections (HSV), CMV, others
Acyclovir, Cidofovir, Famciclovir, Foscarnet, Ganciclovir, Valacyclovir, Valganciclovir
Drugs for Influenza
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian