N-Methylmaleimide
0N-Methylmaleimide is a chemical compound that belongs to the class of organic compounds known as maleimides. It is used in various chemical reactions and has significant applications in biochemistry and pharmacology.
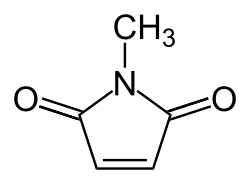
Chemical Structure[edit]
N-Methylmaleimide has a simple structure, consisting of a five-membered ring with two double bonds, one of which is part of an amide group. The other double bond connects the amide group to a methyl group, which gives the compound its name.
Synthesis[edit]
The synthesis of N-Methylmaleimide typically involves the reaction of maleic anhydride with methylamine. This reaction proceeds via nucleophilic acyl substitution, a common mechanism in organic chemistry.
Applications[edit]
N-Methylmaleimide is often used as a reagent in chemical reactions. One of its most notable uses is in the Diels-Alder reaction, a type of cycloaddition that allows for the construction of complex organic structures. In biochemistry, N-Methylmaleimide is used as a protein modifier, specifically targeting cysteine residues.
Safety[edit]
Like many organic compounds, N-Methylmaleimide should be handled with care. It can cause irritation to the skin and eyes, and may be harmful if swallowed or inhaled.
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian

