Iobenguane I-131
What is Iobenguane I-131?[edit]
- Iobenguane I-131 (Azedra) is a radioactive therapeutic agent used for the treatment of pheochromocytoma or paraganglioma.
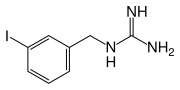
- The drug substance iobenguane I 131 is a substituted benzylguanidine with I 131 in the meta position of the benzene ring.
What are the uses of this medicine?[edit]
- Iobenguane I-131 (Azedra) used for the treatment of adult and pediatric patients 12 years and older with iobenguane scan positive, unresectable, locally advanced or metastatic pheochromocytoma or paraganglioma who require systemic anticancer therapy.
How does this medicine work?[edit]
Azedrais an I 131 labeled iobenguane. Iobenguane is similar in structure to the neurotransmitter norepinephrine (NE) and is subject to the same uptake and accumulation pathways as NE. Pheochromocytoma and paraganglioma (PPGL) are tumors of neural crest origin that express high levels of the NE transporter on their cell surfaces. Following intravenous administration, Azedrais taken up and accumulates within pheochromocytoma and paraganglioma cells, and radiation resulting from radioactive decay of I 131 causes cell death and tumor necrosis.
Who Should Not Use this medicine ?[edit]
- This medicine have no usage limitations.
What drug interactions can this medicine cause?[edit]
Do not administer these drugs until at least 7 days after each Azedradose.
- CNS stimulants or amphetamines (e.g. cocaine, methylphenidate, dextroamphetamine)
- Norepinephrine and dopamine reuptake inhibitors (e.g. phentermine)
- Norepinephrine and serotonin reuptake inhibitors (e.g. tramadol)
- Monoamine oxidase inhibitors (e.g. phenelzine and linezolid)
- Central monoamine depleting drugs (e.g. reserpine)
- Non-select beta adrenergic blocking drugs (e.g. labetalol)
- Alpha agonists or alpha/beta agonists (e.g. pseudoephedrine, phenylephrine, ephedrine, phenylpropanolamine, naphazoline)
- Tricyclic antidepressants or norepinephrine reuptake inhibitors (e.g. amitriptyline, bupropion, duloxetine, mirtazapine, venlafaxine)
- Botanicals that may inhibit reuptake of norepinephrine, serotonin or dopamine (e.g. ephedra, ma huang, St John's Wort, yohimbine)
Is this medicine FDA approved?[edit]
- Iobenguane I 131, marketed under the trade name Azedra, has had a clinical trial as a treatment for malignant, recurrent or unresectable pheochromocytoma and paraganglioma, and the FDA approved it on July 30, 2018.
How should this medicine be used?[edit]
- Verify pregnancy status in females of reproductive potential prior to administering AZEDRA.
- Block thyroid prior to administering AZEDRA.
- Do not administer if platelet count is less than 80,000/mcL or absolute neutrophil count is less than 1,200/mcL.
Recommended dosage: The recommended dosimetric dose is:
- Patients greater than 50 kg: 185 to 222 MBq (5 to 6 mCi)
- Patients 50 kg or less: 3.7 MBq/kg (0.1 mCi/kg)
The recommended therapeutic dose for each of the 2 doses is:
- Patients greater than 62.5 kg: 18,500 MBq (500 mCi)
- Patients 62.5 kg or less: 296 MBq/kg (8 mCi/kg)
Administration:
- Administer AZEDRA intravenously as a dosimetric dose followed by two therapeutic doses administered 90 days apart.
What are the dosage forms and brand names of this medicine?[edit]
This medicine is available in fallowing doasage form:
- As Injection: 555 MBq/mL (15 mCi/ml) at TOC as a clear solution in a single-dose vial.
This medicine is available in fallowing brand namesː
- AZEDRA
What side effects can this medication cause?[edit]
The most common side effects of this medicine include:
- lymphopenia
- neutropenia
- thrombocytopenia
- fatigue
- anemia
- increased international normalized ratio
- nausea
- dizziness
- hypertension
- vomiting
What special precautions should I follow?[edit]
- Azedracontributes to a patient's overall long-term radiation exposure. Long-term cumulative radiation exposure is associated with an increased risk for cancer. Minimize radiation exposure consistent with institutional radiation safety practices and patient management procedures.
- Severe and prolonged myelosuppression occurred during treatment with AZEDRA. Monitor blood cell counts. Withhold and dose reduce Azedraas recommended based on severity of cytopenia.
- Myelodysplastic syndrome (MDS) or acute leukemias were reported in patients who received a therapeutic dose of AZEDRA. The time to development of MDS or acute leukemia ranged from 12 months to 7 years.
- Hypothyroidism was reported in patients who received a therapeutic dose of AZEDRA. Initiate thyroid-blocking medication prior to administration and continue after each dose. Monitor for hypothyroidism and thyroid-stimulating hormone levels before starting Azedraand annually thereafter.
- Elevations in blood pressure may occur. Monitor blood pressure frequently during the first 24 hours after each therapeutic dose.
- Renal Toxicity may occur. Monitor renal function during and after treatment.
- Fatal pneumonitis have occurred. Monitor patients for signs and symptoms of pneumonitis and treat appropriately.
- Based on its mechanism of action, Azedracan cause fetal harm. Advise females and males of reproductive potential of the potential risk to a fetus and to use effective contraception.
- Radiation exposure associated with Azedramay cause infertility in males and females.
What to do in case of emergency/overdose?[edit]
- In case of overdose, call the poison control helpline of your country. In the United States, call 1-800-222-1222.
- Overdose related information is also available online at poisonhelp.org/help.
- In the event that the victim has collapsed, had a seizure, has trouble breathing, or can't be awakened, immediately call emergency services. In the United States, call 911.
Can this medicine be used in pregnancy?[edit]
- Based on its mechanism of action, Azedracan cause fetal harm.
- There are no available data on Azedrause in pregnant women.
Can this medicine be used in children?[edit]
- The safety and effectiveness of Azedrahave not been established in pediatric patients younger than 12 years old with unresectable and iobenguane scan positive, locally advanced or metastatic PPGL which require systemic anticancer therapy.
What are the active and inactive ingredients in this medicine?[edit]
Active ingredient:
- IOBENGUANE I-131
Inactive ingredients:
- SODIUM ASCORBATE
- SODIUM GENTISATE
- WATER
Who manufactures and distributes this medicine?[edit]
Manufactured for:
- Progenics Pharmaceuticals, Inc.
- One World Trade Center, 47th floor, Suite J
- New York, NY
What should I know about storage and disposal of this medication?[edit]
- Store at -70°C (-94°F).
- The shelf life is 6 days post calibration time.
- Discard appropriately at 144 hours.
| Diagnostic radiopharmaceuticals (V09) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
| Therapeutic radiopharmaceuticals (V10) | ||||||||
|---|---|---|---|---|---|---|---|---|
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian