Spinal muscular atrophy 1
Alternate names[edit]
Werdnig-Hoffmann disease; Werdnig Hoffmann disease; Muscular atrophy, infantile; SMA1; SMA, infantile acute form; Proximal spinal muscular atrophy, type 1; Proximal spinal muscular atrophy type 1; SMA type 1; SMA type I; SMA-I
Definition[edit]
Spinal muscular atrophy 1 (SMA1), also known as Werdnig Hoffmann disease, is a genetic neuromuscular disorder that affects the nerve cells that control voluntary muscles (motor neurons).
Epidemiology[edit]
- Spinal muscular atrophy affects 1 per 8,000 to 10,000 people worldwide.
- Spinal muscular atrophy type I is the most common type, accounting for about half of all cases.
Cause[edit]
- SMA1 is caused by changes (pathogenic variants also called mutations) in the SMN1 gene.
- The SMN1 gene provides instructions for making a protein called the survival motor neuron (SMN) protein.
- The SMN protein is one of a group of proteins called the SMN complex, which is important for the maintenance of motor neurons.
- Motor neurons transmit signals from the brain and spinal cord that tell skeletal muscles to tense (contract), which allows the body to move.
- Most people with spinal muscular atrophy are missing a piece of the SMN1 gene, which impairs SMN protein production.
- A shortage of SMN protein leads to motor neuron death, and as a result, signals are not transmitted between the brain and muscles.
- Muscles cannot contract without receiving signals from the brain, so many skeletal muscles become weak and waste away, leading to the signs and symptoms of spinal muscular atrophy.
Inheritance[edit]
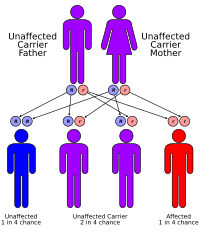
- Spinal muscular atrophy 1 (SMA1) is inherited in an autosomal recessive manner.
- This means that to be affected, a person must have a change (mutation) in both copies of the responsible gene in each cell.
- The parents of an affected person usually each carry one mutated copy of the gene and are referred to as carriers.
- Carriers are typically unaffected and do not have any signs or symptoms of the condition.
When two carriers of an autosomal recessive condition have children, each child has a:
- 25% chance to have the condition
- 50% chance to be an unaffected carrier like each of the parents
- 25% chance to not have the condition and not be a carrier.
An unaffected sibling of a person with SMA1 has a 2/3 chance to be a carrier. Approximately 2% of cases of SMA1 are not inherited from both parents. In these cases, the affected person inherits one mutated copy of the gene from one carrier parent, and has a new mutation that occurs for the first time in the other copy of the gene.
Signs and symptoms[edit]
- Infants with spinal muscular atrophy 1 (SMA1) experience severe weakness before 6 months of age.
- Muscle weakness, lack of motor development and poor muscle tone (hypotonia) are the major features of SMA1.
- Infants with the poorest outlook have problems with breathing and feeding (sucking and/or swallowing).
- Some children develop scoliosis (curvature of the spine) or other skeletal abnormalities.
- Intellectual development is usually normal.
- Affected children are not able to sit up or stand, and the vast majority do not survive past 2 years of age due to respiratory failure.
Diagnosis[edit]
- Genetic testing for spinal muscular atrophy 1 (SMA1) is available.
- Carrier testing for at-risk relatives and prenatal testing for pregnancies at increased risk are possible, if the disease-causing mutations in the family have been identified.
- SMA1 is caused by mutations in the SMN1 gene, and extra copies of the SMN2 gene affect the severity of the condition.
- In some cases, interpreting results of carrier testing for SMA is difficult.
- Approximately 6% of parents of a child with SMA due to deletions in each copy of the gene have normal results of SMN1 dosage testing (carrier testing) for one of two reasons.
- Most people have one copy of SMN1 on each chromosome.
- However, about 4% of carriers have two copies of SMN1 on a single chromosome and a deletion on the other chromosome.
- These carriers with two copies of SMN1 on one chromosome are misdiagnosed as non-carriers by the SMN1 dosage test (they have a false negative test result).
- The second reason is that a new (de novo) deletion on one copy of the SMN1 gene occurs in 2% of people with SMA; in these cases, only one parent is a carrier.
Treatment[edit]
- In December 2016, nusinersen (Spinraza) became the first FDA approved treatment for SMA1.
- Continued treatment with nusinersen is allowing many babies with SMA1 to reach and maintain age appropriate developmental milestones, including sitting, crawling, and walking.
- On average, breathing problems, nutrition problems, and hospital admissions have also decreased.
- However, response to treatment does vary.
- Some babies with SMA1 may not respond to the nusinersen at all or may have medical complications that prevent use of the treatment.
- Other treatments remain supportive.
The medication(s) listed below have been approved by the Food and Drug Administration (FDA) as orphan products for treatment of this condition.
- Risdiplam (Brand name: Evrysdi)was approved for the treatment of spinal muscular atrophy (SMA) in patients 2 months of age and older.
- Nusinersen (Brand name: Spinraza) was approved for the treatment of spinal muscular atrophy in pediatric and adult patients.
- Onasemnogene abeparvovec (Brand name: Zolgensma) was approved for the treatment of pediatric patients less than 2 years of age with spinal muscular atrophy (SMA) with bi-allelic mutations in the survival motor neuron 1 (SMN1) gene.
| Diseases of the nervous system, primarily CNS (G04–G47, 323–349) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| Nucleus diseases | ||||||||
|---|---|---|---|---|---|---|---|---|
see also nucleus
|
NIH genetic and rare disease info[edit]
Spinal muscular atrophy 1 is a rare disease.
| Rare and genetic diseases | ||||||
|---|---|---|---|---|---|---|
|
Rare diseases - Spinal muscular atrophy 1
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian