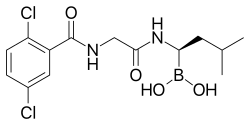
Ixazomib
What is Ixazomib?[edit]
- Ixazomib (Ninlaro) is a proteasome inhibitor used to treat people with multiple myeloma who have received at least one prior therapy.
What are the uses of this medicine?[edit]
- This medicine is used to treat multiple myeloma in combination with the medicines REVLIMID (lenalidomide) and dexamethasone, in people who have received at least one prior treatment for their multiple myeloma.
How does this medicine work?[edit]
- Ixazomib (ix az' oh mib) is an orally available, small molecule inhibitor of the 26S proteasome, the intracellular complex that degrades proteins involved in cell signaling and cell cycle regulation.
- Blocking proteasome activity prevents activation of factors involved in cell growth and resistance to chemotherapy induced apoptosis, thereby leading to cancer cell death.
- Preclinical studies in vitro and in vivo suggested that ixazomib had activity against several hematologic malignancies.
- Clinical trials of the addition of ixazomib to lenalidomide and dexamethasone in patients with multiple myeloma showed improvements in progression free survival.
Who Should Not Use this medicine ?[edit]
- This medicine have no usage limitations.
What drug interactions can this medicine cause?[edit]
- Avoid concomitant administration of Ninlaro with strong CYP3A inducers (such as rifampin, phenytoin, carbamazepine, and St. John's Wort).
Is this medicine FDA approved?[edit]
- It was approved for use in the United States in 2015.
How should this medicine be used?[edit]
Recommended Dosage:
- Ninlaro in combination with lenalidomide and dexamethasone
- The recommended starting dose of Ninlaro is 4 mg administered orally once a week on Days 1, 8, and 15 of a 28-day treatment cycle.
- The recommended starting dose of lenalidomide is 25 mg administered daily on Days 1 through 21 of a 28-day treatment cycle.
- The recommended starting dose of dexamethasone is 40 mg administered on Days 1, 8, 15, and 22 of a 28-day treatment cycle.
Dosage in Patients with Hepatic Impairment
- Reduce the starting dose of Ninlaro to 3 mg in patients with moderate (total bilirubin greater than 1.5-3 × ULN) or severe (total bilirubin greater than 3 × ULN) hepatic impairment.
Dosage in Patients with Renal Impairment
- Reduce the starting dose of Ninlaro to 3 mg in patients with severe renal impairment (creatinine clearance less than 30 mL/min) or end-stage renal disease (ESRD) requiring dialysis.
Administration
- Take Ninlaro exactly as your healthcare provider tells you to take it. Do not change your dose or stop taking Ninlaro without talking to your healthcare provider first.
- Ninlaro is taken in "cycles." Each cycle lasts 4 weeks (28 days).
- The usual dose of Ninlaro is 1 capsule taken 1 time each week, on the same day of the week for the first 3 weeks of each cycle.
- Take each dose of Ninlaro at about the same time of day.
- Take REVLIMID (lenalidomide) and dexamethasone exactly as your healthcare provider tells you to.
- Your healthcare provider will do blood tests during treatment with Ninlaro to check for side effects.
- Your healthcare provider may change your dose or stop Ninlaro, REVLIMID (lenalidomide), or dexamethasone if you have side effects.
- Take Ninlaro at least 1 hour before or at least 2 hours after food.
- On the days that you take both Ninlaro and dexamethasone, do not take Ninlaro and dexamethasone at the same time. Take dexamethasone with food.
- Swallow Ninlaro capsules whole with water. Do not crush, chew or open the capsule.
- Avoid direct contact with the capsule contents. If you accidentally get powder from the Ninlaro capsule on your skin, wash the area well with soap and water. If you accidentally get powder from the Ninlaro capsule in your eyes, flush your eyes well with water.
- If you miss a dose of Ninlaro, or if you are late taking a dose, take the dose as long as the next scheduled dose is more than 3 days (72 hours) away. Do not take a missed dose of Ninlaro if it is within 3 days (72 hours) of your next scheduled dose.
- If you vomit after taking a dose of Ninlaro, do not repeat the dose. Take your next dose of Ninlaro on the next scheduled day and time.
- If you take more Ninlaro than your healthcare provider tells you to take, call your healthcare provider right away or go to the nearest hospital emergency room.
What are the dosage forms and brand names of this medicine?[edit]
This medicine is available in fallowing doasage form: As Capsules: 4 mg, 3 mg, and 2.3 mg
This medicine is available in fallowing brand namesː
- Ninlaro
What side effects can this medication cause?[edit]
The most common side effects of this medicine include:
- diarrhea
- constipation
- thrombocytopenia
- peripheral neuropathy
- nausea
- peripheral edema
- vomiting
- back pain
Ninlaro may cause serious side effects, including:
- Low platelet counts (thrombocytopenia)
- Stomach and intestinal (gastrointestinal) problems
- Nerve problems
- Swelling
- Skin reactions
- Liver problems
What special precautions should I follow?[edit]
- Thrombocytopenia has been reported with Ninlaro. Monitor platelet counts at least monthly during treatment and adjust dosing, as needed.
- Diarrhea, constipation, nausea, and vomiting, have been reported with Ninlaro. Adjust dosing for severe diarrhea, constipation, nausea, and vomiting, as needed.
- The most commonly reported reaction was peripheral sensory neuropathy. Monitor patients for symptoms of peripheral neuropathy and adjust dosing, as needed.
- Peripheral edema may occur. Monitor for fluid retention. Investigate for underlying causes, when appropriate. Adjust dosing, as needed.
- Rash was reported in 19% of patients in the Ninlaro regimen and 11% of patients in the placebo regimen. Monitor patients for rash and adjust dosing, as needed.
- Drug-induced liver injury, hepatocellular injury, hepatic steatosis, hepatitis cholestatic and hepatotoxicity have each been reported. Monitor hepatic enzymes during treatment.
- Ninlaro can cause fetal harm.
- Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception.
What to do in case of emergency/overdose?[edit]
Management of overdosage:
- There is no known specific antidote for Ninlaro overdose.
- In the event of an overdose, monitor the patient for adverse reactions and provide appropriate supportive care.
Can this medicine be used in pregnancy?[edit]
- Ninlaro can cause fetal harm when administered to a pregnant woman.
- There are no human data available regarding the potential effect of Ninlaro on pregnancy or development of the embryo or fetus.
Can this medicine be used in children?[edit]
- Safety and effectiveness have not been established in pediatric patients.
What are the active and inactive ingredients in this medicine?[edit]
- Active ingredient: ixazomib
- Inactive ingredients: microcrystalline cellulose, magnesium stearate, and talc
- Capsule shells: gelatin and titanium dioxide. The 4 mg capsule shell contains red and yellow iron oxide. The 3 mg capsule shell contains black iron oxide. The 2.3 mg capsule shell contains red iron oxide. The printing ink contains shellac, propylene glycol, potassium hydroxide, and black iron oxide.
Who manufactures and distributes this medicine?[edit]
- Distributed and Marketed by:
- Takeda Pharmaceutical Company Limited Cambridge
What should I know about storage and disposal of this medication?[edit]
- Ninlaro may be stored at room temperature. Do not store above 30°C (86°F).
- Do not freeze.
- Store capsules in original packaging until immediately prior to use.
- Ninlaro is cytotoxic.
- Capsules should not be opened or crushed.
- Direct contact with the capsule contents should be avoided.
- In case of capsule breakage, avoid direct contact of capsule contents with the skin or eyes.
- If contact occurs with the skin, wash thoroughly with soap and water.
- If contact occurs with the eyes, flush thoroughly with water.
- Any unused medicinal product or waste material should be disposed in accordance with local requirements.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian