Cladribine
What is Cladribine?[edit]
- Cladribine is a synthetic antineoplastic agent used primarily in the therapy of hairy cell leukemia.
What are the uses of this medicine?[edit]
- Cladribine is used for the treatment of active Hairy Cell Leukemia as defined by anemia, neutropenia, thrombocytopenia or disease-related symptoms.
How does this medicine work?[edit]
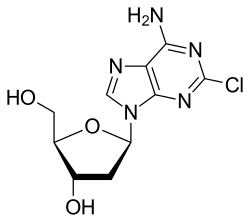
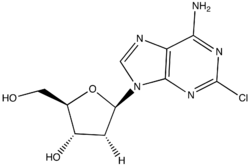
- Cladribine (klad' ri been) is a purine analogue (2-chlorodeoxyadeosine) that is used predominantly in the treatment of hairy cell leukemia.
- Cladribine is a chlorodinated derivative of adenine which is converted intracellularly to the cladribine triphosphate, which is believed to compete with adenine triphosphate in DNA synthesis.
- Cladribine was found to have marked activity against hairy leukemia and was approved for this use in the United States in 1993.
- Cladribine has been used off-label to treat low grade lymphomas and other hematologic malignancies, but its current formal indications are limited to therapy of active hairy cell leukemia.
Who Should Not Use this medicine ?[edit]
This medicine cannot be used in patients:
- who are hypersensitive to this drug or any of its components.
What drug interactions can this medicine cause?[edit]
- There are no known drug interactions with cladribine injection.
- Caution should be exercised if cladribine injection is administered before, after, or in conjunction with other drugs known to cause immunosuppression or myelosuppression.
Is this medicine FDA approved?[edit]
- Cladribine was approved by the FDA in 1993 for hairy cell leukemia as an orphan drug, and was approved in Europe later that year.
How should this medicine be used?[edit]
Recommended dosage:
- The recommended dose and schedule of cladribine injection for active Hairy Cell Leukemia is as a single course given by continuous infusion for seven consecutive days at a dose of 0.09 mg/kg/day.
Administration:
- Cladribine is typically given intravenously daily for 7 days.
What are the dosage forms and brand names of this medicine?[edit]
This medicine is available in fallowing doasage form:
- As injection, USP is available in single-dose vials containing 10 mg (1 mg/mL) of cladribine
This medicine is available in fallowing brand namesː
- CLADRIBINE
What side effects can this medication cause?[edit]
The most common side effects of this medicine include:
- vomiting
- nausea
- stomach pain
- diarrhea
- constipation
- headache
- loss of appetite
- excessive sweating
- skin rash
- pain, redness, swelling, or sores in the place where the medication was injected
cladribine can cause some serious side effects include:
- pale skin
- excessive tiredness
- shortness of breath
- dizziness
- fast heartbeat
What special precautions should I follow?[edit]
- Cladribine injection is a potent antineoplastic agent with potentially significant toxic side effects. It should be administered only under the supervision of a physician experienced with the use of cancer chemotherapeutic agents.
- Serious neurological toxicity (including irreversible paraparesis and quadraparesis) has been reported in patients who received cladribine injection by continuous infusion at high doses.
- Acute nephrotoxicity has been observed with high doses of cladribine injection (four to nine times the recommended dose for Hairy Cell Leukemia), especially when given concomitantly with other nephrotoxic agents/therapies.
- Cladribine has not been associated with serum enzyme elevations during therapy or with instances of clinically apparent acute liver injury with jaundice.
- Due to increased risk of infection in the setting of immunosuppression with chemotherapy including cladribine, it is recommended not to administer live attenuated vaccines to patients receiving cladribine injection.
- Severe bone marrow suppression, including neutropenia, anemia and thrombocytopenia, has been commonly observed in patients treated with cladribine injection, especially at high doses.
- Serious (e.g. respiratory infection, pneumonia and viral skin infections), including fatal infections (e.g., sepsis) were reported.
What to do in case of emergency/overdose?[edit]
Symptoms of overdosage may include:
- irreversible neurologic toxicity (paraparesis/quadriparesis), acute nephrotoxicity, and severe bone marrow suppression resulting in neutropenia, anemia and thrombocytopenia
Management of overdosage:
- There is no known specific antidote to overdosage.
- Treatment of overdosage consists of discontinuation of cladribine, careful observation, and appropriate supportive measures.
- It is not known whether the drug can be removed from the circulation by dialysis or hemofiltration.
Can this medicine be used in pregnancy?[edit]
- Cladribine can cause fetal harm when administered to a pregnant woman.
- Advise females of reproductive potential to use highly effective contraception during treatment with cladribine.
Can this medicine be used in children?[edit]
- Safety and effectiveness in pediatric patients have not been established.
What are the active and inactive ingredients in this medicine?[edit]
Active ingredients include:
- CLADRIBINE
Inactive ingredients include:
- SODIUM CHLORIDE
- PHOSPHORIC ACID
- SODIUM PHOSPHATE, DIBASIC
Who manufactures and distributes this medicine?[edit]
- Packager: Fresenius Kabi USA, LLC
What should I know about storage and disposal of this medication?[edit]
- Store refrigerated 2° to 8°C (36° to 46°F).
- Protect from light (keep in outer carton until time of use).
Alphabetic list of antineoplastic agents - 0-9 - A1 - A2 - A3 - A4 - A5 -A6 - B - C - D - E - F - G - H - I - JK - L - M - NO - PQ - R - S - T - UVW - XYZ
| Antineoplastic Agents | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
* Category
|
| Demyelinating diseases of the central nervous system | ||||||||
|---|---|---|---|---|---|---|---|---|
|
| Merck Serono | ||
|---|---|---|
|
| Immunosuppressive drugs / Immunosuppressants (L04) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian