Crofelemer
What is Crofelemer?[edit]
- Crofelemer (Mytesi) is an anti-diarrheal used to treat noninfectious diarrhea in HIV positive patients on antiretroviral therapy.
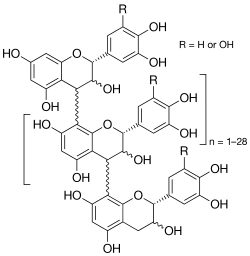
- Crofelemer is an antidiarrheal agent derived from the red sap of the South American plant Croton lechleri.
What are the uses of this medicine?[edit]
- This medicine is used for symptomatic relief of non-infectious diarrhea in adult patients with HIV/AIDS on anti-retroviral therapy.
How does this medicine work?[edit]
- Crofelemer (kroe fel' e mer) is a botanical antidiarrheal agent that is used to treat noninfectious diarrhea in HIV seropositive patients taking antiretroviral medications.
- Crofelemer is derived from the red sap of the South American plant Croton lechleri, which has been used for centuries to treat diarrheal illness.
- The active antidiarrheal product in crofelemer appears to be a large macromolecular, oligomeric proanthocyanidin which has been shown to decrease chloride secretion in the intestine by inhibition of the cystic fibrosis transmembrane conductance regulator (CFTR), as well as calcium-activated chloride channels.
- In large clinical trials, daily therapy with crofelemer was found to decrease watery bowel movements and improve stool consistency in patients with HIV infection receiving antiretroviral therapy, and who had persistent diarrhea that could not be attributed to an infectious cause.
Who Should Not Use this medicine ?[edit]
- This medicine have no usage limitations.
What drug interactions can this medicine cause?[edit]
- Mytesi administration did not have any clinically relevant interaction with nelfinavir, zidovudine, or lamivudine in a drug-drug interaction trial.
Is this medicine FDA approved?[edit]
- Crofelemer was approved for use in the United States in 2012, the first herbal medication to be approved for a specific medical use and first agent approved for therapy of noninfectious diarrhea in HIV positive patients.
How should this medicine be used?[edit]
- Before starting Mytesi, rule out infectious etiologies of diarrhea.
Recommended dosage:
- The recommended adult dosage is 125 mg taken orally twice a day, with or without food.
Administration:
- Do not crush or chew Mytesi tablets.
- Swallow whole.
What are the dosage forms and brand names of this medicine?[edit]
This medicine is available in fallowing doasage form:
- As Delayed-Release Tablets: 125 mg
This medicine is available in fallowing brand namesː
- Mytesi
What side effects can this medication cause?[edit]
The most common side effects of this medicine include:
- upper respiratory tract infection
- bronchitis
- cough
- flatulence
- increased bilirubin
What special precautions should I follow?[edit]
- Before starting Mytesi, rule out infectious etiologies of diarrhea. Mytesi is not indicated for the treatment of infectious diarrhea.
- Instruct patients that Mytesi tablets may be taken with or without food.
- Instruct patients to swallow Mytesi tablets whole and not to crush or chew the tablets.
What to do in case of emergency/overdose?[edit]
- In case of overdose, call the poison control helpline of your country. In the United States, call 1-800-222-1222.
- Overdose related information is also available online at poisonhelp.org/help.
- In the event that the victim has collapsed, had a seizure, has trouble breathing, or can't be awakened, immediately call emergency services. In the United States, call 911.
Can this medicine be used in pregnancy?[edit]
- Crofelemer is minimally absorbed systemically by the oral route of administration and maternal use is not expected to result in fetal exposure to the drug
Can this medicine be used in children?[edit]
- The safety and effectiveness of Mytesi have not been established in pediatric patients.
What are the active and inactive ingredients in this medicine?[edit]
Active ingredient: CROFELEMER
- Inactive ingredients:
CELLULOSE, MICROCRYSTALLINE CROSCARMELLOSE SODIUM SILICON DIOXIDE MAGNESIUM STEARATE ETHYL ACRYLATE TALC TRIETHYL CITRATE XANTHAN GUM TITANIUM DIOXIDE PROPYLPARABEN METHYLPARABEN
Who manufactures and distributes this medicine?[edit]
- Manufactured by Patheon, Inc. for
Napo Pharmaceuticals, Inc., San Francisco, CA
What should I know about storage and disposal of this medication?[edit]
- Store at 20°C-25°C (68°F-77°F); excursions permitted between 15°C-30°C (59°F-86°F).
| Major chemical drug groups | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
| Antidiarrheals, intestinal anti-inflammatory and anti-infective agents (A07) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian