Leukoencephalopathy with vanishing white matter
A rare genetic disorder affecting the central nervous system
| Leukoencephalopathy with vanishing white matter | |
|---|---|
| Autosomal recessive - en.svg | |
| This condition is inherited in an autosomal recessive manner | |
| Synonyms | Childhood ataxia with central nervous system hypomyelinization, Vanishing white matter leukodystrophy, Cree leukoencephalopathy, Vanishing white matter leukodystrophy with ovarian failure, Myelinopathia centralis diffusa
|
| Specialty | Neurology, Medical genetics |
| Symptoms | Ataxia, spasticity, optic atrophy, epilepsy, coma, behavioral changes |
| Complications | Loss of motor function, premature ovarian failure, coma, death |
| Usual onset | Typically childhood, can occur in infancy or adulthood |
| Duration | Progressive |
| Types | Based on age of onset (infantile, childhood, juvenile, adult) |
| Causes | Mutations in EIF2B1, EIF2B2, EIF2B3, EIF2B4, or EIF2B5 genes |
| Risk factors | Inherited autosomal recessive trait |
| Diagnosis | MRI, genetic testing, neurological examination |
| Differential diagnosis | Multiple sclerosis, other leukodystrophies |
| Prevention | None |
| Treatment | Supportive care, symptom management, avoidance of stressors |
| Medication | Anticonvulsants for seizures, supportive treatments |
| Prognosis | Poor, especially with early onset |
| Frequency | Rare |
| Deaths | Common in severe forms |
Leukoencephalopathy with vanishing white matter (VWM), also known as vanishing white matter disease, is a rare genetic disorder that primarily affects the central nervous system (CNS). It is characterized by the progressive loss of white matter in the brain, leading to a variety of neurological symptoms.
Pathophysiology[edit]
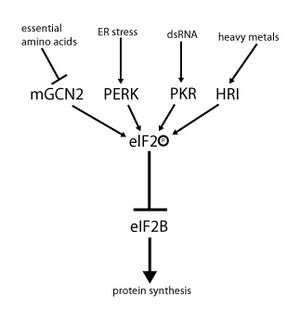
Leukoencephalopathy with vanishing white matter is caused by mutations in any of the five genes encoding the subunits of the eukaryotic translation initiation factor 2B (eIF2B). These genes are EIF2B1, EIF2B2, EIF2B3, EIF2B4, and EIF2B5. The eIF2B complex is crucial for the regulation of protein synthesis, particularly under conditions of cellular stress. Mutations in these genes disrupt normal protein synthesis, leading to the degeneration of white matter in the brain.
The disease is characterized by the progressive disappearance of white matter, which is replaced by cerebrospinal fluid. This process is visible on magnetic resonance imaging (MRI) scans, which show a characteristic pattern of white matter loss.
Clinical Presentation[edit]
The clinical presentation of VWM can vary widely, but it typically includes:
Symptoms often begin in early childhood, but the age of onset can range from infancy to adulthood. The disease is progressive, and stressors such as fever, minor head trauma, or infections can exacerbate the symptoms.
Diagnosis[edit]
Diagnosis of leukoencephalopathy with vanishing white matter is based on clinical evaluation, MRI findings, and genetic testing. MRI scans reveal the characteristic pattern of white matter loss, while genetic testing can confirm mutations in the eIF2B genes.
Management[edit]
There is currently no cure for VWM, and treatment is primarily supportive. Management focuses on alleviating symptoms and preventing complications. This may include:
- Physical therapy to manage spasticity and improve mobility
- Anticonvulsant medications to control seizures
- Supportive care for cognitive and developmental issues
Avoidance of stressors that can exacerbate the disease is also crucial.
Prognosis[edit]
The prognosis for individuals with leukoencephalopathy with vanishing white matter varies depending on the age of onset and the severity of the disease. Early-onset cases tend to have a more rapid progression and poorer prognosis, while later-onset cases may progress more slowly.
Related pages[edit]
| Diseases of the nervous system, primarily CNS (G04–G47, 323–349) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| Disorders of translation and posttranslational modification | ||||
|---|---|---|---|---|
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian