Miltefosine
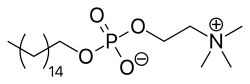
Miltefosine is a pharmaceutical agent primarily prescribed for the treatment of leishmaniasis and specific free-living amoeba infections. Marketed under the brand name Impavido among other labels, this drug has revolutionized the treatment of some challenging parasitic diseases, particularly within the tropical disease domain.
Indications[edit]
Miltefosine has shown therapeutic benefits against a spectrum of parasitic infections:
Leishmaniasis: The drug combats all three clinical manifestations of this condition:
Free-Living Amoeba Infections:
- Naegleria fowleri: A rare, but often fatal, brain infection[3].
- Balamuthia mandrillaris: An amoeba that affects the skin and the central nervous system.
In certain scenarios or for particular patient demographics, Miltefosine might be prescribed alongside other antiparasitic treatments, such as liposomal amphotericin B or paromomycin.
Administration[edit]
Administered orally, Miltefosine offers a preferable alternative to the often cumbersome intravenous or intramuscular treatments generally prescribed for parasitic afflictions.
Mechanism of Action[edit]
While ongoing research is still demystifying its exact mechanism, existing evidence suggests that Miltefosine disrupts the cellular metabolism of parasites, halting their growth and proliferation[4].
Adverse Effects[edit]
Like many drugs, Miltefosine has associated side effects that can range from mild to severe:
Common:
- Vomiting
- Abdominal pain
- Fever
- Headaches
- Impaired kidney function
Severe:
- Stevens-Johnson syndrome: A severe dermatological reaction.
- Thrombocytopenia: A condition characterized by reduced blood platelet count[5].
Contraindications[edit]
Pregnant individuals should refrain from Miltefosine due to documented teratogenic effects on the fetus[6]. Lactating mothers should also avoid this medication due to the potential risk it poses to infants.
Conclusion[edit]
Miltefosine heralds a new era in the treatment of leishmaniasis and certain amoebic infections. As the medical landscape continues its evolution, the balance between this drug's therapeutic potential and its associated risks becomes ever more critical, ensuring both patient safety and effective outcomes.
References[edit]
- ↑ Sundar, S., & Chakravarty, J. (2015). Leishmaniasis: An update of current pharmacotherapy. Expert Opinion on Pharmacotherapy, 16(2), 237-252.
- ↑ Chappuis, F., Sundar, S., Hailu, A., Ghalib, H., Rijal, S., Peeling, R. W., ... & Boelaert, M. (2007). Visceral leishmaniasis: What are the needs for diagnosis, treatment, and control? Nature Reviews Microbiology, 5(11), S7-S16.
- ↑ Grace, E., Asbill, S., & Virga, K. (2015). Naegleria fowleri: Pathogenesis, diagnosis, and treatment options. Antimicrobial Agents and Chemotherapy, 59(11), 6677-6681.
- ↑ Dorlo, T. P., Balasegaram, M., Beijnen, J. H., & de Vries, P. J. (2012). Miltefosine: A review of its pharmacology and therapeutic efficacy in the treatment of leishmaniasis. Journal of Antimicrobial Chemotherapy, 67(11), 2576-2597.
- ↑ Rijal, S., Ostyn, B., Uranw, S., Rai, K., Bhattarai, N. R., Dorlo, T. P., ... & Boelaert, M. (2013). Increasing failure of miltefosine in the treatment of kala-azar in Nepal and the potential role of parasite drug resistance, reinfection, or noncompliance. Clinical Infectious Diseases, 56(11), 1530-1538.
- ↑ Dorlo, T. P., Huitema, A. D., Beijnen, J. H., & de Vries, P. J. (2012). Optimal dosing of miltefosine in children and adults with visceral leishmaniasis. Antimicrobial Agents and Chemotherapy, 56(7), 3864-3872.
| Antiviral drugs: antiretroviral drugs used against HIV (primarily J05) | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| Antiparasitics directed at excavata parasites (P01) | ||||
|---|---|---|---|---|
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
- Antiprotozoal agents
- Antifungals
- Quaternary ammonium compounds
- Antiretroviral drugs
- Antineoplastic drugs
- Zwitterionic surfactants
- Protein kinase inhibitors
- Embryotoxicants
- Fetotoxicants
- Drugs acting on the blood and blood forming organs
- Antivirals
- German inventions
- World Health Organization essential medicines
- RTT