Elagolix
What is Elagolix?[edit]
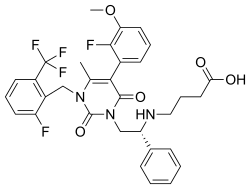
- Elagolix (Orilissa) is a gonadotropin-releasing hormone (GnRH) receptor antagonist used to treat moderate to severe pain associated with endometriosis.
What are the uses of this medicine?[edit]
- This medicine is used to treat moderate to severe pain associated with endometriosis.
Limitations of Use
- Limit the duration of use based on the dose and coexisting condition.
How does this medicine work?[edit]
- An orally bioavailable, second-generation, non-peptide based, small molecule compound and selective gonadotropin-releasing hormone (GnRH; LHRH) receptor antagonist, with potential hormone production inhibitory activity.
- Upon oral administration, elagolix competes with GnRH for receptor binding and inhibits GnRH receptor signaling in the anterior pituitary gland.
- This inhibits the secretion of luteinizing hormone (LH) and follicle-stimulating hormone (FSH).
- In males, the inhibition of LH secretion prevents the release of testosterone.
- In women, inhibition of FSH and LH prevents the production of estrogen by the ovaries.
- Inhibition of GnRH signaling may treat or prevent symptoms of sex hormone-dependent disease states.
Who Should Not Use this medicine ?[edit]
This medicine cannot be used in patients:
- Who are pregnant
- With known osteoporosis because of the risk of further bone loss
- With severe hepatic impairment
- Taking inhibitors of organic anion transporting polypeptide (OATP)1B1 (a hepatic uptake transporter)
- With known hypersensitivity reaction to Orilissa or any of its inactive components
What drug interactions can this medicine cause?[edit]
- Concomitant use of Orilissa 200 mg twice daily and strong CYP3A inhibitors for more than 1 month is not recommended. Limit concomitant use of Orilissa 150 mg once daily and strong CYP3A inhibitors to 6 months.
- Co-administration of Orilissa with strong CYP3A inducers may decrease elagolix plasma concentrations and may result in a decrease of the therapeutic effects of Orilissa.
- Concomitant use of Orilissa 200 mg twice daily and rifampin is not recommended. Limit concomitant use of Orilissa 150 mg once daily and rifampin to 6 months.
- The effect of concomitant use of P-gp inhibitors or inducers on the pharmacokinetics of Orilissa is unknown.
Is this medicine FDA approved?[edit]
- It was approved for use in the United States in 2018.
How should this medicine be used?[edit]
- Exclude pregnancy before starting Orilissa or start Orilissa within 7 days from the onset of menses.
Recommended Dosage Normal liver function or mild hepatic impairment:
- 150 mg once daily for up to 24 months or 200 mg twice daily for up to 6 months.
Moderate hepatic impairment:
- 150 mg once daily for up to 6 months.
Administration
- Take Orilissa exactly as your healthcare provider tells you to take it.
- Your healthcare provider will give you a pregnancy test before you start taking Orilissa or will have you start taking Orilissa within 7 days after you start your period.
If your healthcare provider prescribes:
- Orilissa 150 mg (a pink tablet), take it 1 time each day
- Orilissa 200 mg (an orange tablet), take it 2 times each day
- Take Orilissa at about the same time each day with or without food.
- If you take too much Orilissa, call your healthcare provider or go to the nearest hospital emergency room right away.
If you miss a dose of Orilissa:
- 150 mg (1 time each day), take it as soon as you remember as long as it is on the same day. Do not take more than 1 tablet each day.
- 200 mg (2 times each day), take it as soon as you remember as long as it is on the same day. Do not take more than 2 tablets each day.
What are the dosage forms and brand names of this medicine?[edit]
This medicine is available in fallowing doasage form:
- As Oral tablets: 150 mg and 200 mg
This medicine is available in fallowing brand namesː
- Orilissa
What side effects can this medication cause?[edit]
The most common side effects of this medicine include:
- hot flashes and night sweats
- headache
- nausea
- difficulty sleeping
- absence of periods
- anxiety
- joint pain
- depression
- mood changes
Orilissa can cause serious side effects including:
- suicidal thoughts, suicidal behavior, and worsening of mood
- abnormal liver tests
signs and symptoms of liver problems:
- yellowing of the skin or the whites of the eyes (jaundice)
- dark amber-colored urine
- feeling tired (fatigue or exhaustion)
- nausea and vomiting
- generalized swelling
- right upper stomach area (abdomen) pain
- bruising easily
What special precautions should I follow?[edit]
- Inform patients about the risk of bone loss. Advise patients that supplementary calcium and vitamin D may be beneficial if dietary intake of calcium and vitamin D is not adequate.
- Advise women that Orilissa may delay the recognition of pregnancy because it may reduce the amount, intensity, or duration of menstrual bleeding. Advise patients to use effective non-hormonal contraception while taking Orilissa and to discontinue Orilissa if pregnancy is confirmed.
- Advise patients that suicidal ideation and exacerbation of mood disorders may occur with Orilissa use.
- Advise patients to promptly seek medical attention in case of signs or symptoms that may reflect liver injury, such as jaundice.
- Use non-hormonal contraception during treatment and for 28 days after discontinuing Orilissa. Coadministration of Orilissa 200 mg twice daily with an estrogen-containing contraceptive is not recommended because of the potential for increased estrogen-associated risks. Coadministration of Orilissa with an estrogen-containing contraceptive may reduce the efficacy of Orilissa. Coadministration with progestin-containing oral contraceptives may reduce the efficacy of the contraceptive.
What to do in case of emergency/overdose?[edit]
Management for overdosage:
- In case of overdose, monitor the patient for any signs or symptoms of adverse reactions and initiate appropriate symptomatic treatment, as needed.
Can this medicine be used in pregnancy?[edit]
- Use of Orilissa is contraindicated in pregnant women.
- Exposure to Orilissa early in pregnancy may increase the risk of early pregnancy loss.
- Discontinue Orilissa if pregnancy occurs during treatment.
- There is a pregnancy registry that monitors outcomes in women who become pregnant while treated with Orilissa. Pregnant patients should be encouraged to enroll by calling 1-833-782-7241.
Can this medicine be used in children?[edit]
- Safety and effectiveness of Orilissa in pediatric patients have not been established.
What are the active and inactive ingredients in this medicine?[edit]
- Active ingredient: elagolix
- Inactive ingredients 150 mg tablets: mannitol, sodium carbonate monohydrate, pregelatinized starch, povidone, magnesium stearate, polyvinyl alcohol, titanium dioxide, polyethylene glycol, talc, and carmine high tint.
- Inactive ingredients 200 mg tablets: mannitol, sodium carbonate monohydrate, pregelatinized starch, povidone, magnesium stearate, polyvinyl alcohol, titanium dioxide, polyethylene glycol, talc, and iron oxide red.
Who manufactures and distributes this medicine?[edit]
Manufactured by AbbVie Inc. North Chicago
What should I know about storage and disposal of this medication?[edit]
- Store Orilissa between 36°F to 86°F (2°C to 30°C).
- Do not keep medicine that is out of date or that you no longer need.
- Throw away (dispose of) unused medicines through community take-back disposal programs when available. If no community take-back disposal program is available go to www.fda.gov/drugdisposal for more information on how to dispose of Orilissa the right way.
- Do not flush Orilissa down the toilet.
- Keep Orilissa and all medicines out of the reach of children.
| Hypothalamic-pituitary hormones and analogues (H01) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| GnRH and gonadotropin receptor modulators | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian