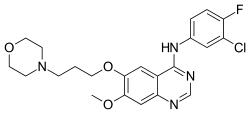
Gefitinib
What is Gefitinib?[edit]
- Gefitinib (Iressa) is a tyrosine kinase inhibitor used in the therapy of non-small cell lung cancer.
What are the uses of this medicine?[edit]
Gefitinib (Iressa) used to treat people with non-small cell lung cancer (NSCLC) that has spread to other parts of the body and:
- that have certain types of abnormal epidermal growth factor receptor (EGFR) genes, and
- who have not had previous treatment for cancer
Limitation of Use:
- It is not known if Iressa is safe and effective in people with NSCLC that have other types of EGFR genes.
How does this medicine work?[edit]
- Gefitinib (ge fi' ti nib) is a selective inhibitor of the tyrosine kinase receptor of epidermal growth factor (EGFR), which is often mutated and over expressed in cancer cells, particularly non-small cell lung cancer and some forms of breast cancer.
- The mutated EGF tyrosine kinase receptor is constitutively expressed which causes unregulated cell growth and proliferation.
- By inhibition of this growth factor receptor, gefitinib blocks the intracellular Ras signaling transduction cascade, which results in inhibition of the malignant cell growth.
- Highest rates of response to gefitinib are seen in patients with activating mutations of EGFR in the tumor tissue.
Who Should Not Use this medicine ?[edit]
Limitation of Use:
- It is not known if Iressa is safe and effective in people with NSCLC that have other types of EGFR genes.
What drug interactions can this medicine cause?[edit]
Tell your doctor and pharmacist what prescription and nonprescription medications, vitamins, and nutritional supplements you are taking or plan to take. Be sure to mention any of the following:
- anticoagulants ('blood thinners') such as warfarin (Coumadin, Jantoven)
- antifungals such as itraconazole (Onmel, Sporanox) and ketoconazole (Nizoral)
- metoprolol (Lopressor, Toprol XL, in Dutoprol); phenytoin (Dilantin, Phenytek)
- tricyclic antidepressants such as imipramine (Tofranil) and amitriptyline
- If you take a proton pump inhibitor (PPI), H2 blocker, or an antacid medicine take antacid at least 6 hours before or 6 hours after taking gefitinib and take proton pump inhibitor at least 12 hours before or at least 12 hours after taking gefitinib.
Is this medicine FDA approved?[edit]
- Gefitinib received approval for use in the United States in 2009 for the treatment of advanced non-small cell lung cancer after failure of other therapies, but has been available in Japan since 2002.
How should this medicine be used?[edit]
Recommended dosage:
- Recommended dose is 250 mg orally, once daily with or without food.
Strong CYP3A4 Inducers:
- Increase Iressa to 500 mg daily in the absence of severe adverse drug reaction, and resume Iressa at 250 mg seven days after discontinuation of the strong CYP3A4 inducer.
Administration:
- Take Iressa 1 time each day.
- You can take Iressa with or without food.
- If you miss a dose of Iressa, take it as soon as you remember.
- If it is less than 12 hours until your next dose, skip the missed dose.
- Take your next dose at your regular time.
If you cannot swallow Iressa tablets whole:
- place your dose of Iressa in a container with 4 to 8 ounces of water and stir for about 15 minutes
- drink the mixture right away
- place another 4 to 8 ounces of water in the same container, and drink it right away
What are the dosage forms and brand names of this medicine?[edit]
This medicine is available in fallowing doasage form:
- As Tablets: 250 mg
This medicine is available in fallowing brand namesː
- IRESSA
What side effects can this medication cause?[edit]
The most common side effects of this medicine include:
- skin reactions
- diarrhea
IRESSA may cause serious side effects, including:
- lung or breathing problems
- liver problems
- perforation
- diarrhea
- eye problems
- skin reactions
- IRESSA may cause fertility problems in females.
What special precautions should I follow?[edit]
- Interstitial lung disease (ILD) occurred in patients taking Iressa. Withhold Iressa for worsening of respiratory symptoms. Discontinue Iressa if ILD is confirmed.
- Gefitinib therapy is associated with transient elevations in serum aminotransferase levels and rare instances of clinically apparent acute liver injury. Obtain periodic liver function testing. WithholdIressa for Grade 2 or higher for ALT and/or AST elevations. Discontinue for severe hepatic impairment.
- Gastrointestinal perforation occurred in three Iressa-treated patients. Discontinue Iressa for gastrointestinal perforation.
- Grade 3 or 4 diarrhea occurred in Iressa-treated patients. Withhold Iressa for Grade 3 or higher diarrhea.
- Ocular disorders occurred in the 2462Iressa-treated patients. Withhold Iressa for signs and symptoms of severe or worsening ocular disorders including keratitis. Discontinue for persistent ulcerative keratitis.
- Bullous conditions including toxic epidermal necrolysis, Stevens Johnson syndrome and erythema multiforme have been reported from treatment with Iressa. Withhold Iressa for Grade 3 or higher skin reactions or exfoliative conditions.
- Based on its mechanism of action and data from animal reproduction studies Iressa can cause fetal harm when administered to a pregnant woman. Advise of potential risk to a fetus and use of effective contraception.
What to do in case of emergency/overdose?[edit]
Symptoms of overdosage may include:
- Adverse events were mostly mild to moderate in severity, and were consistent with the known safety profile of Iressa.
Management of overdosage:
- In the event of suspected overdose, interrupt Iressa, institute supportive care, and observe until clinical stabilization.
- There are no specific measures/treatments that should be taken following Iressa overdosing.
Can this medicine be used in pregnancy?[edit]
- Based on its mechanism of action and animal data, IRESSA can cause fetal harm when administered to a pregnant woman.
- Advise pregnant women of the potential hazard to a fetus or potential risk for loss of the pregnancy.
Can this medicine be used in children?[edit]
- The safety and effectiveness of Iressa in pediatric patients have not been established.
What are the active and inactive ingredients in this medicine?[edit]
- Active ingredient: gefitinib
- Inactive ingredients: lactose monohydrate, microcrystalline cellulose, croscarmellose sodium, povidone, sodium lauryl sulfate, magnesium stearate
- Tablet coating contains: hypromellose, polyethylene glycol 300, titanium dioxide, yellow iron oxide and red iron oxide.
Who manufactures and distributes this medicine?[edit]
Distributed by:
- AstraZeneca Pharmaceuticals LP
- Wilmington, DE
What should I know about storage and disposal of this medication?[edit]
- Store Iressa at room temperature between 68˚F to 77˚F (20˚C to 25˚C)
Alphabetic list of antineoplastic agents - 0-9 - A1 - A2 - A3 - A4 - A5 -A6 - B - C - D - E - F - G - H - I - JK - L - M - NO - PQ - R - S - T - UVW - XYZ
| Antineoplastic Agents | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
* Category
|
| Growth factor receptor modulators | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| AstraZeneca | ||||||
|---|---|---|---|---|---|---|
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian