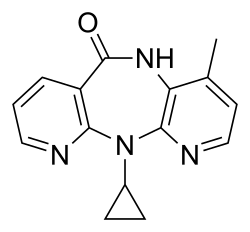
Nevirapine
Non-nucleoside reverse transcriptase inhibitor used in HIV treatment
| Nevirapine | |
|---|---|
| Trade names | Viramune
|
| Chemical nomenclature | |
| IUPAC name | 11-cyclopropyl-4-methyl-5,11-dihydro-6H- dipyrido[3,2-b:2′,3′-e][1,4]diazepin-6-one
|
| Routes | By mouth
|
| Bioavailability | 93% ± 9% |
| Metabolism | Liver |
| Half-life | 45 hours |
| Excretion | Primarily via kidney (<6% as parent drug) and bile duct (<5% as parent drug) |
| Legal status | Prescription only |
| Identifiers | |
| CAS Number | 129618-40-2 |
| PubChem | 4463 |
| DrugBank | DB00238 |
| ChemSpider | 4308 |
| UNII | 99DK7FVK1H |
| KEGG | D00435 |
| ChEMBL | 57 |
| Chemical data
| |
| Chemical formula | C15H14N4O1
|
Overview[edit]
Nevirapine is a non-nucleoside reverse transcriptase inhibitor (NNRTI) used in combination with other antiretroviral drugs to treat human immunodeficiency virus (HIV) infection and acquired immune deficiency syndrome (AIDS). It functions by directly inhibiting the HIV reverse transcriptase enzyme, preventing the replication of viral RNA.
Nevirapine is commonly included in highly active antiretroviral therapy (HAART) and is known for its efficacy in reducing viral load in HIV-positive individuals.
Mechanism of Action[edit]
Nevirapine inhibits HIV reverse transcriptase, the enzyme necessary for viral RNA conversion into DNA, a key step in HIV replication. Unlike nucleoside reverse transcriptase inhibitors (NRTIs), nevirapine does not require intracellular activation. Instead, it binds directly to the enzyme, causing a conformational change that disrupts its function, preventing viral replication.
Indications[edit]
Nevirapine is indicated for:
- HIV-1 infection in combination with other antiretroviral agents.
- Prevention of mother-to-child transmission (PMTCT) during childbirth.
Dosage and Administration[edit]
Nevirapine is available in tablet (200 mg) and oral suspension forms. Standard dosing includes:
- Initial dose: 200 mg once daily for 14 days.
- Maintenance dose: 200 mg twice daily OR 400 mg once daily (extended-release).
- Pediatric dosing: Adjusted based on weight and age.
Nevirapine is always used in combination with other antiretroviral therapy (ART) agents to reduce the risk of HIV resistance.
Side Effects[edit]
Common side effects include:
- Rash (up to 20% of patients)
- Nausea and vomiting
- Fatigue
- Fever
- Headache
Severe adverse effects:
- Hepatotoxicity: Nevirapine is linked to severe liver toxicity, particularly in patients with pre-existing liver conditions.
- Stevens-Johnson Syndrome (SJS) and Toxic Epidermal Necrolysis (TEN): Severe skin reactions requiring immediate medical attention.
Contraindications and Precautions[edit]
Nevirapine should not be used in:
- Patients with severe hepatic impairment.
- Individuals with prior hypersensitivity reactions to the drug.
- Women with CD4+ counts >250 cells/mm³ and men >400 cells/mm³ due to increased risk of liver toxicity.
Drug Interactions[edit]
Nevirapine is a known CYP3A4 inducer, which can decrease plasma concentrations of certain drugs, including:
- Oral contraceptives (may reduce effectiveness).
- Protease inhibitors (PIs) like lopinavir/ritonavir.
- Warfarin and other anticoagulants.
It is advised to monitor drug levels when used with other CYP3A4-metabolized drugs.
Pharmacokinetics[edit]
- Absorption: Well-absorbed orally (93% bioavailability).
- Distribution: Widely distributed with high tissue penetration.
- Metabolism: Primarily hepatic via the CYP3A4 enzyme system.
- Elimination: Half-life of ~45 hours, primarily excreted via urine.
Special Populations[edit]
- Pregnancy: Classified as Category B3 in Australia. Used cautiously in pregnant women for PMTCT.
- Pediatrics: Dose adjustments required for children based on weight.
- Renal impairment: Minimal renal excretion; dose adjustment typically not required.
Hepatotoxicity[edit]
Nevirapine has a well-established link to hepatotoxicity, ranging from mild liver enzyme elevations to severe liver failure. Risk factors include:
- Pre-existing liver disease (e.g., Hepatitis B or C).
- Higher baseline CD4+ counts in women (>250) and men (>400).
- Concomitant hepatotoxic medications.
Availability and Market Status[edit]
Nevirapine is widely available as a generic medication and is part of the World Health Organization’s (WHO) List of Essential Medicines.
Brand names include:
- Viramune (Boehringer Ingelheim)
- Various generic formulations
Alternatives[edit]
Nevirapine is part of the NNRTI class of antiretrovirals. Other NNRTI alternatives include:
Patients experiencing severe side effects or drug resistance may be switched to integrase inhibitors (INSTIs) or protease inhibitors (PIs).
See Also[edit]
- Highly active antiretroviral therapy (HAART)
- HIV/AIDS treatment
- Reverse transcriptase inhibitors
- Drug-induced liver injury
External Links[edit]
| Antiviral drugs: antiretroviral drugs used against HIV (primarily J05) | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian