Idoxifene
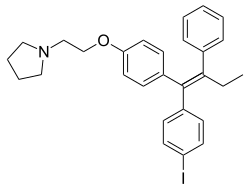
Idoxifene: An Investigated SERM[edit]
Idoxifene (also known by its International Nonproprietary Name (INN), US Adopted Name (USAN), and British Approved Name (BAN)), and previously identified under developmental code names CB-7432 and SB-223030, belongs to the category of nonsteroidal Selective Estrogen Receptor Modulators (SERM). It is structurally related to the triphenylethylene group and is alternatively termed pyrrolidino-4-iodotamoxifen. While it was being considered for clinical use, especially in the treatment of breast cancer and postmenopausal osteoporosis, it never progressed to commercialization.
Chemical and Pharmacological Background[edit]
- Structural Classification: Idoxifene is structurally a member of the triphenylethylene group, which associates it with other well-known SERMs. Its chemical structure is analogous to tamoxifen, with a pyrrolidino-4-iodo substitution.
- Mode of Action: As a SERM, Idoxifene modulates the estrogen receptor's activity, functioning either as an agonist or antagonist based on the target tissue[1].
Clinical Development[edit]
Idoxifene underwent extensive clinical trials to evaluate its therapeutic potential in two primary medical conditions:
- Breast Cancer: Idoxifene reached phase II clinical trials for its efficacy in treating breast cancer. Breast cancer cells often exhibit an over-expression of estrogen receptors, and modulating these receptors can offer therapeutic benefits[2].
- Postmenopausal Osteoporosis: Recognizing the influence of estrogen on bone mineral density, Idoxifene was explored in phase III clinical trials for its potential in treating osteoporosis in postmenopausal women[3].
Unfortunately, Idoxifene's development was halted in 1999, as it failed to demonstrate sufficient therapeutic efficacy in both of these conditions.
Challenges in Development[edit]
While the precise reasons for the discontinuation of Idoxifene's development aren't publicized in full detail, the generic challenges faced in the development of SERMs include:
- Efficacy: Achieving a balance where a SERM can suppress estrogenic activity in one tissue (like the breast) while promoting it in another (like bone) is intricate.
- Safety: All SERMs carry potential side effects, some of which may not be apparent until advanced stages of clinical trials.
Conclusion[edit]
Idoxifene, despite its initial promise as a potent SERM, did not meet the desired endpoints in clinical trials, reinforcing the inherent challenges in developing efficacious and safe therapeutic agents in this category. Its study, however, adds to the body of knowledge on SERMs, potentially aiding in the development of more effective drugs in the future.
References[edit]
- ↑ Maximov, P. Y., Lee, T. M., & Jordan, V. C. (2013). The discovery and development of selective estrogen receptor modulators (SERMs) for clinical practice. Current Clinical Pharmacology, 8(2), 135-155.
- ↑ Smith, C. L., & O'Malley, B. W. (2004). Coregulator function: a key to understanding tissue specificity of selective receptor modulators. Endocrine Reviews, 25(1), 45-71.
- ↑ Riggs, B. L., & Hartmann, L. C. (2003). Selective estrogen-receptor modulators — mechanisms of action and application to clinical practice. New England Journal of Medicine, 348(7), 618-629.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian